Running Head: Exercise-Induced Desaturation in Early COPD Rehab
Funding Support: This work was supported by the Ricerca Corrente Funding scheme of the Ministry of Health, Italy.
Date of Acceptance: February 13, 2026 | Published Online Date: February 25, 2026
Abbreviations: 6MWD=6-minute walking distance; 6MWT=6-minute walking test; AECOPD=acute exacerbation of chronic obstructive pulmonary disease; ANOVA=analysis of variance; BMI=body mass index; CAT=COPD Assessment Test; CIRS=Cumulative Illness Rating Scale; CIRS-CCI=Cumulative Illness Rating Scale with Comorbidity Complexity Index; COPD=chronic obstructive pulmonary disease; EID=exercise-induced desaturation; FEV1=forced expiratory volume in 1 second; FEV₁ %pred=forced expiratory volume in 1 second percentage predicted; FiO2=fraction of inspired oxygen; FVC=forced vital capacity; HR=heart rate; LoS=length of stay; LTOT=long-term oxygen therapy; mMRC=modified Medical Research Council dyspnea scale; PaO2=positive pressure of oxygen; PR=pulmonary rehabilitation; SpO2=peripheral oxygen saturation
Citation: Paneroni M, Spinello L, Salvi B, et al. Prevalence of exercise-induced desaturation among COPD patients enrolled in early inpatient pulmonary rehabilitation. Chronic Obstr Pulm Dis. 2026; 13(2): 136-146. doi: http://doi.org/10.15326/jcopdf.2025.0724
Introduction
Acute exacerbations of chronic obstructive pulmonary disease (AECOPDs) are sudden, severe worsening of respiratory symptoms that require a change in a person's usual treatment.1 The severity of these episodes is classified by the level of medical intervention needed: mild (no steroids or antibiotics), moderate (requiring steroids and/or antibiotics, often managed outpatient), and severe (requiring hospitalization or an emergency department visit).1
AECOPDs can have significant long-term negative consequences, including a faster decline in lung function, an increased risk of future exacerbations, and a higher chance of developing chronic hypoxemia (low blood oxygen).1 For those individuals with severe hypoxemia during the acute phase of an exacerbation, the risk of adverse outcomes increases.2
AECOPD episodes can also lead to heart problems, increased inflammation, and worsening cognitive and mental health issues, along with a rapid decline in physical strength and independence.1,3
Even when their oxygen levels are normal at rest, many people with stable chronic obstructive pulmonary disease (COPD) experience a significant drop in oxygen during physical activity.4 This is known as exercise-induced desaturation (EID). Several cohort studies have quantified the prevalence of EID in populations with stable COPD without respiratory failure. Reports indicate that this prevalence ranges from approximately 21% to over 60%, depending on the sample and the definitions used.5-9
Importantly, EID has been recognized as a prognostic factor linked to increased mortality and higher risk of exacerbations in COPD patients, as highlighted by García-Talavera et al.10
Most available evidence comes from studies on stable outpatients11 while much less is known about the immediate postexacerbation period. This leaves some uncertainty about how frequently desaturation occurs during this fragile phase. Research shows that this is often linked to underlying structural lung damage and can lead to shortness of breath, fatigue, and a reluctance to exercise.12-14
Therefore, there is a lack of information on how quickly oxygen levels recover after a moderate to severe AECOPD. While some studies show gradual improvement over several weeks, research suggests that many patients can safely discontinue oxygen therapy after being reassessed, highlighting the importance of proper follow-up after hospitalization to avoid unnecessary long-term oxygen prescriptions.12-15
Early pulmonary rehabilitation (PR) is a crucial step in recovery after a moderate or severe exacerbation. Even during a PR stay, some patients may develop relevant oxygen desaturation with exercise with and without oxygen use, despite having normal resting values. This can contribute to tiredness and may worsen deconditioning over time. The 6-minute walking test (6MWT) is widely used in rehabilitation. It is beneficial for identifying patients who are desaturated on exertion and might benefit from supplemental oxygen during exercise.
To our knowledge, no prevalence studies have been performed on EID in patients admitted for PR after AECOPDs. The primary purpose of our study was to measure the prevalence of EID in patients at admission to hospital rehabilitation recovering from AECOPDs with and without need for oxygen therapy at rest.
The secondary aim was to compare clinical and functional status, as well as the response to the walk test performed at admission, between subgroups of combined oxygen need and EID presence. Response to the walk test was assessed by measuring performance (meters walked), desaturation time course, and differences in dyspnea, fatigue, and heart rate.
Materials and Methods
This multicenter, retrospective study was approved by the Ethical Committee of Istituti Clinici Scientifici Maugeri, IRCCS, Pavia, Italy (EC Lombardia 6 Prot. 18390/25; 31 March 2025). At admission, patients gave their written informed consent for the scientific use of their data. As a retrospective analysis, the study was not registered.
Patients
This study was conducted on the automated integrated health care record database of patients with COPD, consecutively admitted for in-hospital pulmonary rehabilitation after an AECOPD between August 1, 2022 and November 30, 2024, at the hospitals of Istituti Clinici Scientifici Maugeri (Lumezzane and Ginosa), referral institutions for pulmonary rehabilitation, diagnosis, and care of chronic patients.
Inclusion Criteria
All patients who had been diagnosed with COPD by spirometry, in accordance with the Global initiative for chronic Obstructive Lung Disease (GOLD) guidelines,1 and who were admitted to an inpatient PR program, were enrolled. Patients were transferred to our Institutions within 15 days following an AECOPD, which required either acute hospital care or home management by the general practitioner. At admission, all the patients were in sufficient stable conditions as follows: absence of worsening in symptoms (i.e., no change in cough and/or sputum beyond day-to-day variability which would have been sufficient to warrant a change in the latest management prescribed at discharge from the hospital or prescribed at home by a general practitioner), and stability in blood gas values (e.g., no respiratory acidosis). All patients received regular treatment according to current guidelines for their disease stage.1
Exclusion criteria were a diagnosis other than COPD, no availability of spirometry to perform a correct COPD diagnosis, orthopedic, neurological, or oncological comorbidities, or subacute or unstable conditions, and the presence of significant dyspnea at rest.
Patients were divided into 2 groups based on their condition at admission: those breathing room air at rest (group 1) and those receiving supplemental oxygen at rest (group 2), which included both patients who had a long-term oxygen therapy (LTOT) prescription before AECOPD and patients who started oxygen therapy during exacerbation and still needed an oxygen supply. The presence or absence of LTOT before exacerbation was not used as an inclusion or exclusion criterion. In this study, the type of oxygen therapy prescribed during the rehabilitation program did not necessarily correspond to the home prescription at the time of discharge. Although traditional criteria for an LTOT prescription (positive pressure of oxygen [PaO2] ≤55mmHg or PaO2 <60mmHg with evidence of cor pulmonale or polycythaemia) were indirectly applied, it should be emphasized that these criteria have not been specifically validated in unstable patients in the postexacerbation setting.16
Indication for the inpatient program was: patient too fragile or unstable to participate safely in outpatient rehabilitation, recent severe exacerbation or hospitalization with persistent symptoms/functional impairment, severe resting hypoxemia or frequent exercise-induced desaturation requiring close monitoring and oxygen titration, severe physical deconditioning or frailty (unable to walk or transfer independently), high risk of falls or mobility limitations (needs physiotherapist supervision, e.g., wheelchair-bound), significant comorbidities that require medical supervision (e.g., advanced heart failure, pulmonary hypertension), need for multidisciplinary intervention in a controlled setting, including nutritional support, psychosocial care, and complex therapy coordination, lack of a safe home environment, or inability to access outpatient/tele-rehabilitation.
Measurements
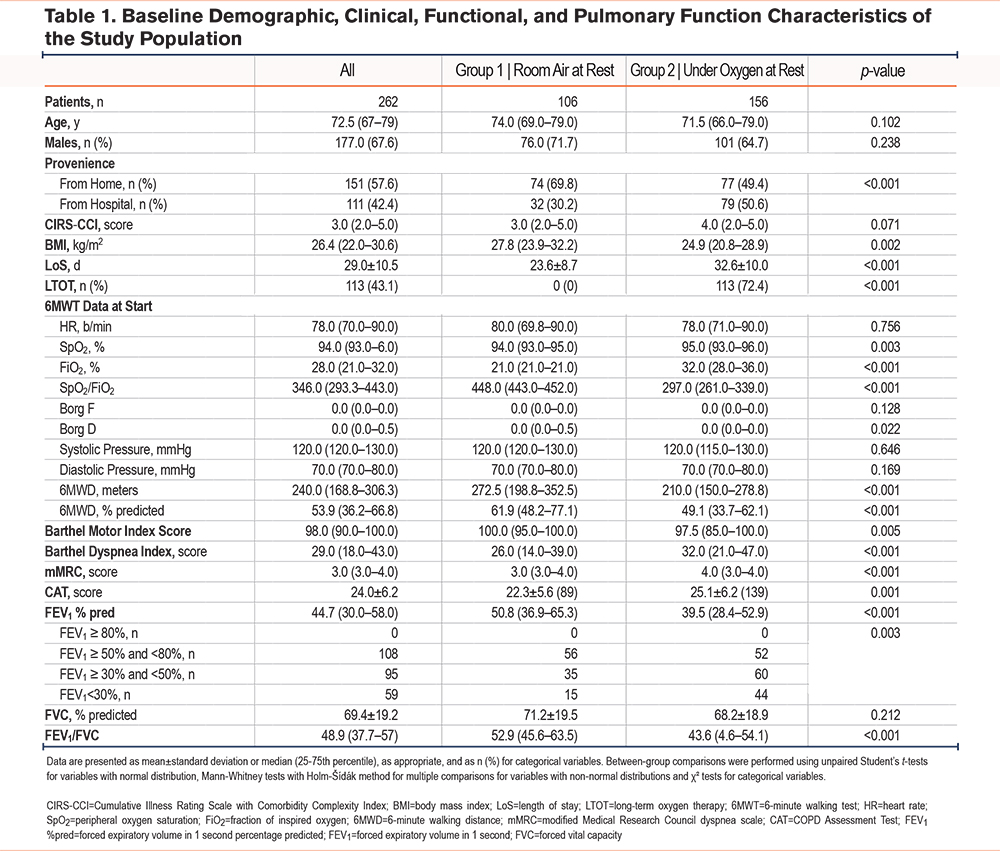
Anthropometrics, demographics, length of stay in our centers, comorbidities with the Cumulative Illness Rating Scale (CIRS) with the Comorbidity Complexity Index,17 dynamic lung volumes (forced expiratory volume in 1 second [FEV1] percentage predicted [%pred], forced vital capacity [FVC] %pred, FEV1/FVC) according to standards,18 using the predicted values of Quanjer19 were measured; severity FEV1 classes were also calculated.
Motor disability by the Barthel index,20 dyspnea by the Italian version of the Barthel index dyspnea,21 functional dyspnea by the modified Medical Research Council (mMRC) scale,22 and the symptom impact by the COPD Assessment Test (CAT) score23 were collected.
Exercise tolerance (meters and percentage of predicted) was performed by the 6MWT24 under oxygen supply according to their usual LTOT prescription or new prescription at discharge from the acute hospital (group 2) or under room air at rest (group 1), maintaining a peripheral oxygen saturation (SpO2) above 92% throughout the test, with the following baseline measures: oxygen saturation (SpO2), fraction of inspired oxygen (FiO2), SpO2/FiO2, and systolic/diastolic arterial pressure. All patients on oxygen used continuous-flow oxygen delivery systems. At baseline and at the end of the test, we evaluated Borg dyspnea and fatigue, while oxygen saturation (SpO2) and heart rate were continuously measured during the entire test, using a validated portable pulse oximeter (NONIN Model 8500 Handheld Pulse Oximeter, Nonin Medical Inc.; Plymouth, Minnesota).
EID was defined as a decrease in SpO2 from baseline to nadir of≥4 percentage points during the 6MWT combined with a nadir SpO2<90% according to several authors.4,5,25
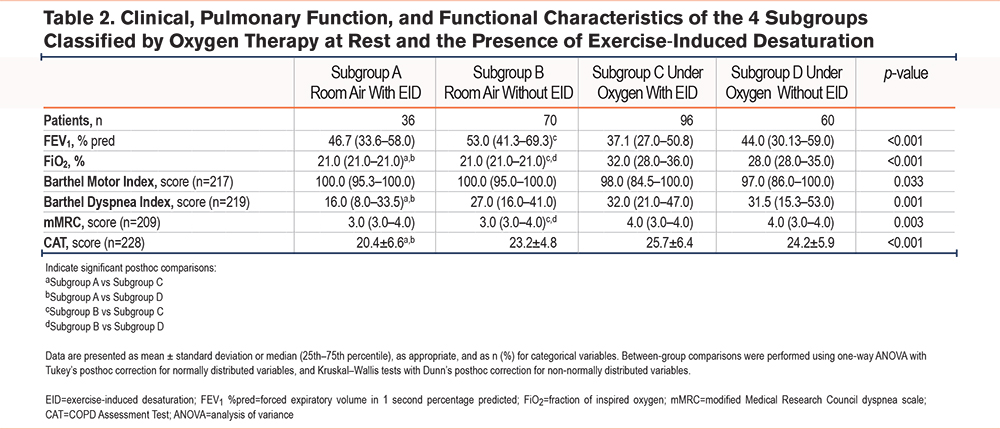
Patients were then classified into 4 subgroups according to the EID presence: Subgroup A=Room Air with EID; Subgroup B=Room Air without EID; Subgroup C=Under Oxygen with EID; Subgroup D=Under Oxygen without EID.
Program
The inpatient multidisciplinary program included optimizing drug therapy, education, nutritional programs, and psychosocial counseling when appropriate, as well as at least 22 sessions over 3–4 weeks of supervised incremental exercise training. The total daily time duration of activities was 2–3 hours/day.26
Statistical Analysis
All analyses were performed using GraphPad Prism version 9.0.0 (GraphPad Software; San Diego, California) and Microsoft Excel (Microsoft Corp.; Redmond, Washington). Continuous variables were reported as mean ± standard deviation for normally distributed data or as median (25th–75th percentile) for non-normally distributed data, while categorical variables were expressed as absolute numbers and percentages.
Normality of distribution was assessed using the Shapiro–Wilk test. Parametric tests were used for variables with a normal distribution, whereas nonparametric tests were applied otherwise. For comparisons between 2 groups, unpaired 2-tailed Student’s t-tests were used for normally distributed variables and Mann–Whitney U tests for non-normally distributed ones.
For comparisons among more than 2 groups, 1-way analysis of variance (ANOVA) followed by Tukey’s posthoc test was used for normally distributed variables, and Kruskal–Wallis tests with Dunn’s multiple comparison correction were used for non-normally distributed variables.
Categorical variables were compared using Pearson’s χ2 test, or Fisher’s exact test for 2×2 tables with small expected frequencies.
A p-value <0.05 was considered statistically significant, and exact p-values are reported wherever relevant.
Results
Out of 1200 patients admitted to institutions in the study period, 262 patients with all inclusion criteria participated: 106 patients breathing room air at rest and 156 patients receiving supplemental oxygen at rest.
The median age of the study population was 72.5 years (25th–75th percentile 67–79), and two-thirds of the participants were men (177 out of 262, 67.6%). The 2 groups did not differ in age or sex (p=0.102 and p=0.238, respectively). A total of 151 of 262 patients (57.6%) came from home, while 111 (42.4%) were from the hospital. Patients breathing room air at rest were admitted more frequently from home (69.8%) compared with those receiving supplemental oxygen at rest (49.4%), (p<0.001). Among the 262 patients, 106 breathed room air at rest; among those who required oxygen at rest (156 patients), 27.6% received short-term oxygen therapy, and 72.4% were on LTOT. Compared with those breathing room air, patients who needed oxygen at rest had a more compromised profile: a significantly lower body mass index (p=0.002), longer length of stay (p<0.001), and lower Barthel motor scores (p=0.005), and higher Barthel dyspnea (p<0.001), mMRC (p<0.001), and CAT (p=0.001) scores. They also had more impaired pulmonary function (lower FEV₁ percentage, p<0.001; lower FEV₁/FVC, p<0.001) and a shorter 6-minute walk distance (6MWD) (p<0.001), while baseline heart rate and Borg scores were comparable between groups (p =0.756 and p=0.128–0.022, respectively). Table 1 summarizes the main baseline characteristics.
A total of 132 patients out of 262 (50.4%) admitted patients presented EID; EID occurred in 36 out of 106 patients (33.9%) who breathed room air and 96 out of 156 patients (61.5%) who received supplemental oxygen at rest (Figure 1). The difference between groups was statistically significant (χ2=19.2, df=1, p<0.001).

Within the group under oxygen at rest, no statistical difference was found regarding EID between the LTOT group and the non-LTOT group (65.5% versus 51.2%, p=0.140).
For subgroup analyses (Figure 1), participants were divided into 4 groups: Room Air with EID (n=36, group A), Room Air without EID (n=70, group B), Oxygen with EID (n=96, group C), and Oxygen without EID (n=60, group D). Table 2 presents the baseline clinical, functional, and pulmonary characteristics of the 4 subgroups. Patients already on oxygen (groups C and D) showed a clearly more compromised profile compared with those breathing room air (groups A and B). FiO2 was higher in patients who experienced EID during the 6MWT. FEV1%pred differed significantly among subgroups (Kruskal–Wallis p<0.001), with posthoc contrasts showing lower values in the oxygen groups compared with room air, particularly between Room Air with EID and Under Oxygen with EID (p<0.001). Functional capacity, measured by the Barthel motor score, was lower among patients on oxygen (Kruskal–Wallis p=0.033), although pairwise comparisons were not significant (all Dunn’s p>0.05). Disability related to dyspnea was greater, as shown by higher Barthel-dyspnea scores (Kruskal–Wallis p=0.001), and significant posthoc differences between Room Air with EID versus Under Oxygen with EID (p=0.001). The perception of symptoms and disease burden was also worse, with a higher mMRC (Kruskal–Wallis p=0.003), and significant contrasts were observed between Room Air without EID and Under Oxygen without EID (p=0.043). CAT scores (1-way ANOVA p<0.001), with Tukey posthoc showing differences for Room Air with EID versus Under Oxygen with EID (p<0.001).
The 6MWD is shown to have significant overall subgroup differences in Figure 2 (Kruskal–Wallis p=0.014); Dunn’s multiple comparison test identified a significant difference between Room Air with EID versus Under Oxygen with EID (Dunn’s p<0.002) and between Room Air without EID versus Under Oxygen without EID (Dunn’s p<0.033). Patients receiving supplemental oxygen at rest walked significantly shorter distances on the 6MWT compared to those breathing room air, regardless of the presence of EID. Within each oxygen group (room air or oxygen), there were no significant differences in walking distance between patients with EID and those without EID.
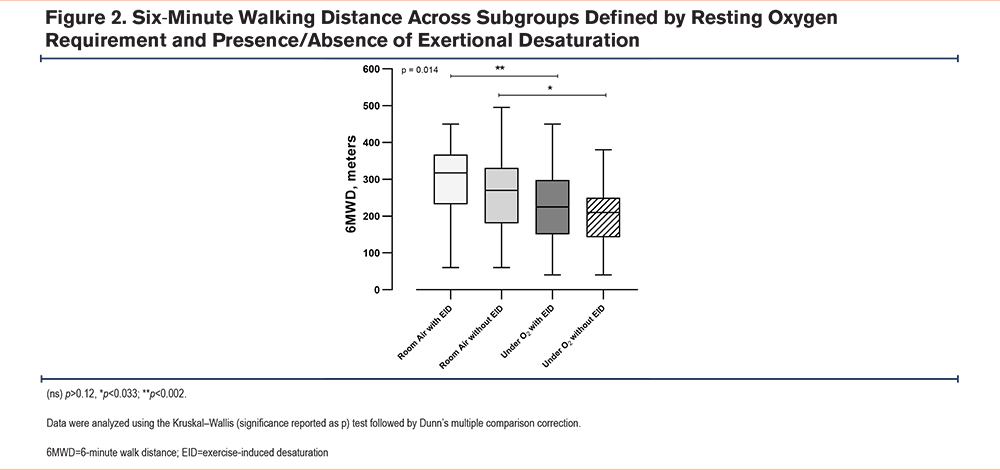
Figure 3 illustrates the comparison between subgroups in terms of their responses to the walking test. Changes in Borg fatigue (Figure 3A) and Borg dyspnea (Figure 3B) scores, indicating the perceived exertion, did not show significant differences among the 4 subgroups.
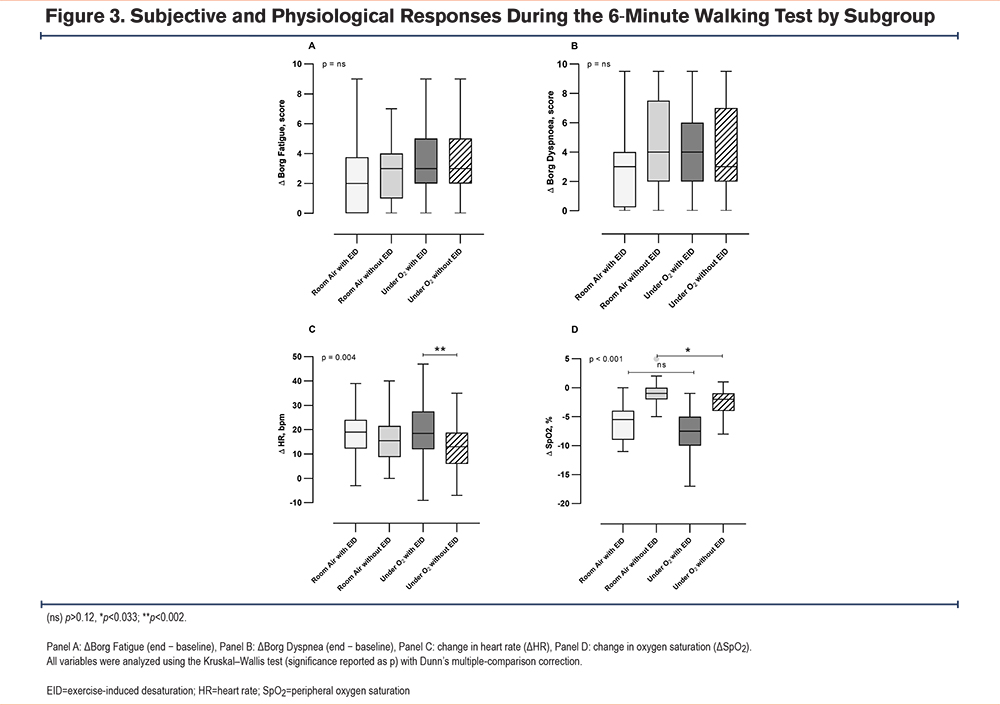
The change in heart rate (Figure 3C) showed an overall difference among subgroups (Kruskal–Wallis p=0.004). Posthoc analysis with Dunn's correction revealed a significant difference specifically between the Under Oxygen with EID and Under Oxygen wihout EID subgroups (p=0.003), while no other pairwise comparisons were significant.
The change in SpO2 (Figure 3D) was highly significant in pairwise contrasts (Kruskal–Wallis, p <0.001). Post hoc analysis with Dunn’s correction highlights a significant difference (p<0.033) between the non-EID groups.
Patients in groups A (n=36) and C (n=96), who experience EID, were trained in the rehabilitation setting using oxygen as needed in order to maintain SpO2 above 90% during exercise.
Considering the provenience before admission, the prevalence of EID did not vary significantly between patients using oxygen therapy coming from home (68.8%) and the hospital (54.4%) (p=0.0685), nor in patients breathing room air coming from home (31.1%) or the hospital (40.6%) (p=0.3431).
Also, exercise capacity as measured by the 6MWD did not show clear differences between the subgroups regarding provenience: patients breathing room air coming from home walked 282.6 ± 106.2 meters and patients from the hospital walked 258.0 ± 91.1 meters (p=0.2566), while patients breathing oxygen coming from home walked 224.7 ± 96.1 meters and patients from the hospital walked 214.3 ± 85.4 meters (P=0.4757).
Discussion
Our study observed that EID using a combined index, as a drop in oxygen saturation (SpO2) and a decrease of at least 4% from baseline with a nadir <90%, is common among individuals with COPD undergoing rehabilitation post-AECOPD, representing 50% of the cases. This condition occurs twice as often (61.5%) in patients who are already using oxygen at rest.
These findings need to be interpreted in consideration of the broader consequences of severe exacerbations. Hospitalization for a severe AECOPD leads to accelerated lung function decline, increased future exacerbations, triggered acute cardiac events, increased systemic inflammation, worsened cognitive decline, anxiety, depression, and worsened rapid deconditioning, muscle weakness, and exercise tolerance.1,3
Among negative consequences after an AECOPD, studies indicate that patients may present severe oxygen desaturation with a higher risk of adverse outcomes.2 However, we still know relatively little about how hypoxemia recovers after a moderate-to-severe exacerbation. Recent research suggests that recovery patterns can be highly variable. For example, Oliveira et al12 observed gradual improvements in SpO2 and FEV₁ over the first 45 days postexacerbation, highlighting the diverse trajectories among patients.
A particular condition of hypoxemia is EID. However, differences in how EID is defined and the types of patients studied also strongly affect reported prevalence. Yang et al,4 for instance, found that about two-thirds of stable COPD patients (62.1%) experienced drops in oxygen levels during exercise. This desaturation was linked to lower carbon monoxide diffusion capacity, higher CAT scores, and more extensive emphysema on computed tomography scans.4 These results suggest that structural lung changes and impaired gas exchange can cause significant oxygen desaturation during activity, even in patients with normal resting oxygen levels.4
An AECOPD has meaningful clinical consequences, often leading to respiratory failure, reported in roughly 41% of hospitalized patients, which can result from ventilation-perfusion mismatch, air trapping, and inspiratory muscle overload, all contributing to hypercapnia and impaired oxygenation.27-29
On the other hand, many studies have indicated that various clinical and functional patient characteristics can significantly impact the outcomes of the 6MWT. This includes factors such as the distance walked, levels of O2 desaturation, and the perception of dyspnea.30
Within this context, the 50% EID prevalence during the 6MWT observed in our study becomes particularly relevant; the high rate of EID among patients already on oxygen (61.5%) mirrors the prevalence reported in cohorts of more severe or stable COPD5,6,8 suggesting that many postacute patients retain gas-exchange impairment similar to chronically severe disease. This observation is consistent with the understanding that individuals with more severe COPD and impaired gas exchange are at higher risk for desaturation during exertion.31
Conversely, the 34% prevalence among room-air patients, while lower, still represents a clinically relevant minority who may be at risk of activity-related hypoxaemia. Prior studies have precisely linked EID to accelerated FEV₁ decline, increased mortality, and an increased risk of exacerbation.7,32
Beyond desaturation, our subgroup analysis revealed broader clinical differences. Patients receiving oxygen, regardless of whether they experienced EID, showed a more limited exercise capacity and reduced lung function compared with those breathing room air. These patients also showed poorer functional status and reported more disability due to breathlessness, along with a higher overall symptom burden. This suggests that individuals admitted on oxygen represent a particularly vulnerable group, in which EID is just one aspect of a broader and more severe clinical profile.
Despite the oxygen therapy aimed at preventing desaturation, many patients still exhibited EID, indicating a need for ongoing monitoring and individualized oxygen titration.This result can be justified, on the one hand, by the greater disability and severity of patients admitted with oxygen at rest (Table 1), but could also suggest that the usual oxygen prescription used in the chronic phase is no longer adequate after severe AECOPDs, or that the methods for prescribing oxygen therapy during exertion are not adequately standardized and are subject to many variables, such as the patient's condition, presence of comorbidities, the intensity and quality of physical activity performed, and the correct use of the prescribed oxygen device.
LTOT combined with exercise rehabilitation is more effective than exercise alone in improving exercise capacity, pulmonary function, and quality of life in COPD patients32 while the evidence to support the use of oxygen compared to room air during exercise training in EID patients is lacking,33 and further investigations specifically focused on postexacerbation rehabilitation populations should be considered. As expected, exercise tolerance in patients without oxygen at rest was higher than in patients with oxygen. Of note, in both groups, desaturators were able to walk more meters in the 6MWT than nondesaturators, even without reaching statistical significance. A possible explanation for this could lie in the fact that, precisely because they perform better, some COPD patients push themselves harder during exercise and experience desaturation.
Interestingly, symptom assessment alone (dyspnea and fatigue) as heart rate response often fails to detect EID and does not appear to correlate directly with the level of desaturation (Figure 3); indeed, in daily life, many desaturation episodes go unnoticed without continuous oximetry during functional testing such as the 6MWT.
How rehabilitation and natural recovery interact with this phenomenon is still uncertain. The extent to which the observed phenomenon can be modified by a rehabilitation program or the extent to which the time factor can allow for natural improvement remains a subject for future studies. Additionally,33 using functional respiratory imaging showed ventilation–perfusion matching improvements during recovery, offering further mechanistic insights into recovery patterns.
Several studies evaluated postdischarge oxygen needs, where many patients were eligible for oxygen discontinuation.11,13,34 These critical results documented meaningful changes in oxygenation and therapy needs upon reassessment, reinforcing the importance of structured follow-up to avoid unnecessary long-term oxygen prescriptions.
We have used a combined criterion (SpO2<90% or ΔSpO2≥4%) to test EID, a criterion also applied in recent prospective protocols, reflecting current consensus on clinically meaningful EID.35 Among alternative EID criteria, nadir-centered rules detect a larger number of desaturators while maintaining good agreement with end-of-test criteria; using a nadir threshold increases sensitivity for detecting transient but clinically relevant desaturation during the 6MWT.4
Study Limitations and Strengths
Our study has limitations. Like all retrospective studies, it suffers from missing data, such as in CIRS, Barthel scales, CAT, and mMRC. Pre-exacerbation values of oxygenation, oxygen liters need, and exercise capacity were not available. Consequently, it is unknown whether the COPD severity, level of oxygenation, and exercise capacity before the exacerbations were equal or not.
However, we are confident that our findings of increased EID in patients receiving oxygen at rest support the importance of early identification of vulnerable individuals and personalized strategies for rehabilitation, monitoring, and oxygen titration. Objective measures obtained during the 6MWT, particularly continuous SpO2 monitoring, provided critical information not captured by symptom scores alone.
Another important limitation of this study is the definition of EID adopted, based on a≥4% reduction in oxygen saturation and a nadir of SpO2 below 90%. As no universally accepted definition exists, slight variations are present across studies.35,36 The definition used may have influenced the estimated prevalence and limited the comparability of results.
Conclusions
Among patients admitted to inpatient pulmonary rehabilitation after an AECOPD, EID (defined as a fall in SpO2 to <90% and a≥4% drop from baseline) was common and more frequent in those already on oxygen therapy at rest. Patients receiving oxygen therapy also had worse clinical and functional status and walked shorter distances on the 6MWT. Regular reassessment of oxygen prescriptions, along with systematic monitoring of EID, could help identify those at higher risk and allow rehabilitation programs to be better tailored to individual needs. Further prospective studies are needed to confirm the current findings and to clarify how EID evolves, whether it affects long-term outcomes, and whether interventions guided by its detection can improve patient prognosis.
Acknowledgments
Author contributions: MP was responsible for the conceptualization of the manuscript. BS, CS, and AD were in charge of the investigation. LS, BS, CS, and AD were responsible for data curation. MP conducted the formal analysis. MVitacca was in charge of the methodology. MP and MVitacca supervised. LS, BS, CS, and AD contributed to the visualization of the manuscript. MVitacca wrote the original draft. LS, BS, MP, CS, AD, MVenturelli, and MVitacca reviewed and edited the manuscript.
Other acknowledgements: The authors thank Laura Comini and Adriana Olivares for their criticism and technical assistance.
The authors would like to acknowledge the support of the Italian Ministry of University and Research (MUR) through the National Recovery and Resilience Plan (PNRR), funded by the European Union – NextGenerationEU, Mission 4 “Education and Research”, Component 1. This work was carried out within the framework of project CUP B31I24000510004.
Declaration of Interest
All authors report no conflicts of interest or financial support relating to this paper.
Data availability statement: The data that support the findings of this study are available from the corresponding author upon reasonable request.
IA use: Open Evidence has been used as an additional tool in literature search.