Running Head: Linguistic and Cultural Validation of the CERT
Funding Support: This study was funded by GSK (study number 222091). GSK funded the article publication fee.
Date of Acceptance: March 2, 2026 | Published Online Date: March 9, 2026
Abbreviations: CERT=COPD Exacerbation Recognition Tool; CERT-J=COPD Exacerbation Recognition Tool–Japan; COPD=chronic obstructive pulmonary disease; FDA=Food and Drug Administration; GAAPP=Global Allergy and Airways Patient Platform; LVC=linguistic validation consultant; PACE=Predictors of Acute COPD Exacerbation study
Citation: Gloeckl R, Tal-Singer R, Deussen P, et al. COPD exacerbation recognition tool: translation, linguistic, and cross-cultural validation. Chronic Obstr Pulm Dis. 2026; 13(2): 147-157. doi: http://doi.org/10.15326/jcopdf.2025.0745
Online Supplemental Material: Read Online Supplemental Material (390KB)
Note: A summary of these results was presented at the European Respiratory Society 2025 Congress in Amsterdam, Netherlands and was then encored at the Korean Academy of Tuberculosis and Respiratory Diseases International Conference 2025 in Seoul, Korea. A summary of these results was also encored at the 30th French-Language Pulmonary Congress in Lille, France in January 2026.
Introduction
Chronic obstructive pulmonary disease (COPD) is one of the leading causes of morbidity and mortality worldwide and has a considerable economic burden.1,2 COPD exacerbations have been shown to increase health care resource utilization and costs and to negatively impact patients’ quality of life.3 Exacerbation frequency is an important determinant of lung function decline4; as exacerbation frequency and severity increases, the risk of future exacerbations and mortality also increases.5 Despite this, patients experience difficulty recognizing an exacerbation,6 and approximately 40% of COPD exacerbations are not immediately reported by patients.7-10 Additionally, patients have been shown to report exacerbations in some instances, but not in others.10 Opportunities for treatment changes can, therefore, be missed, and this may have a subsequent impact on health status.11,12
The COPD Exacerbation Recognition Tool (CERT) was first developed in China to help patients recognize when they are experiencing a COPD exacerbation and to encourage them to seek prompt medical assessment.13 The development study aimed to identify the words and phrases used by patients in China to describe symptoms associated with the onset of a COPD exacerbation.13 The key symptoms identified were worsening cough, increased sputum volume, shortness of breath, labored breathing, and limitation of activity.13 Moderate or severe worsening in at least 2 of these symptoms relative to the patient’s usual state had good sensitivity (91.8%) and specificity (100%) for an exacerbation.13 A recent study conducted in China in a different population of patients with COPD has shown that the CERT has high sensitivity (92.4%) and specificity (72.1%) for moderate and severe exacerbations.14 A second developmental study took place in Japan, independently of the development of the CERT in China.15 Using similar methodology to the original CERT study, this Japan-specific CERT identified the same symptoms as those listed in the original CERT, with the addition of a sixth symptom (change in sputum color) that was added at the request of physician advisors.15
Another recent study, conducted in Germany, used a German translation of the CERT items in the form of a daily diary to determine whether the tool could identify the onset of a COPD exacerbation.16 Patient responses to the CERT when it was used in this way were shown to predict exacerbations one day before the clinical diagnosis was made and showed good sensitivity and specificity (70% and 91%, respectively).16
To improve the accessibility of the CERT, it has been translated into over 60 languages, taking care to ensure that the translations were conceptually equivalent to the original, comparable across languages, culturally relevant to the context of the target country, and easily understood by patients. To this aim, we conducted a cross-cultural and linguistic validation of 46 CERT translations. In this paper we focus the results on 5 translation languages from 5 different regions that represent a broad range of cultures and geographic scope: Indonesian, Korean, Norwegian, Spanish for use in Latin America, and Universal Arabic (Modern Standard).
Materials and Methods
COPD Exacerbation Recognition Tool Translation
The original version of the CERT, written in Simplified Chinese (Mandarin), was translated into English.13 The translations and cultural and linguistic validation of the CERT employed rigorous, certified (International Organization for Standardization-17100) methodology and were performed in accordance with Food and Drug Administration (FDA) guidance17 and industry standards.18,19 This ensured that the translation was conceptually equivalent to the original English translation and harmonized with other translations. A total of 46 CERT translations across 25 countries and 6 continents were linguistically and culturally validated in this study (Table 1). An overview of the CERT translation process is shown in Figure 1.

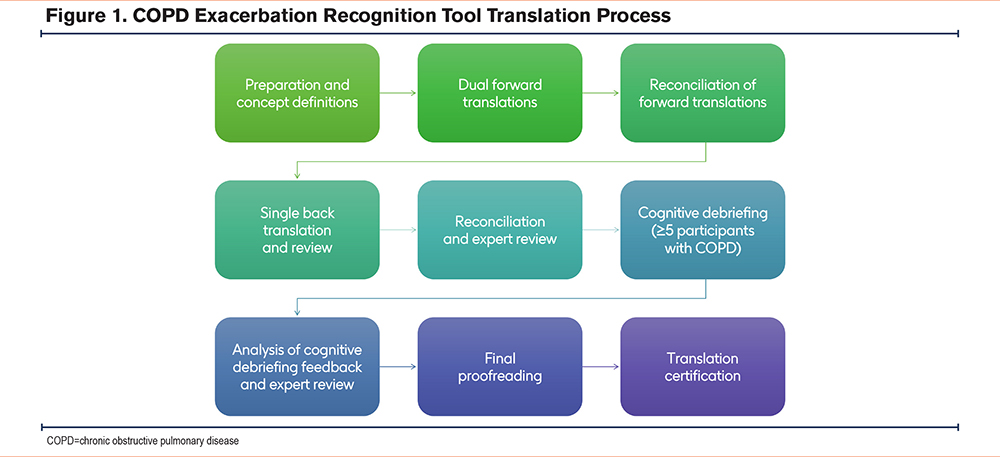
Preparation and Concept Definitions:
An overview of the preparation of CERT translations and concept definitions is included in the online supplement. The concept elicitation guide is presented in Supplemental Table 1 in the online supplement.
Dual Forward Translations:
Two forward translations were developed by independent translators who were native speakers of the target language, fluent in English, and experienced in the translation of clinical outcome assessment measures.
Reconciliation of Forward Translations:
A linguistic validation consultant (LVC) reconciled the 2 forward translations into a third translation by comparing, choosing, and combining the most appropriate translation and/or wording from the 2 initial translations, while ensuring that the reconciled version was conceptually equivalent to the English translation.
Single Back Translation and Review:
A back translation of the reconciled translation was provided by independent linguists in the source language. In each case, the back translator was fluent in the target language and English, and had no prior knowledge of the original CERT. The back translations were reviewed against a U.S.-English translation of the Simplified Chinese source CERT by the project manager, who directed any queries to the LVC. The LVC then refined the translation to correct any discrepancies or errors based on recommendations and comments from the back review.
Reconciliation and Expert Review:
The translation was submitted to the expert reviewers, who reviewed the decisions that had been made throughout the translation process up to this point. Any comments or questions that arose from this review were discussed between the project manager and the LVC until they were resolved.
Cognitive Debriefing:
Linguistic validation was achieved through a series of individual interviews with at least 5 participants with COPD, conducted by the LVC. To be included in this study, participants needed to be diagnosed with COPD, aged ≥18 years (ideally ≥45 years as COPD is more prevalent in mature individuals20), and native speakers of the target language. We aimed to include participants of a mix of genders (ideally 2 males and 3 females, due to expected increase in prevalence of COPD in females compared with males21), educational backgrounds, and socioeconomic statuses. An overview of the interview process is included in the online supplement.
Analysis of Cognitive Debriefing Feedback and Expert Review:
An overview of the analysis of cognitive debriefing feedback and expert review is included in the online supplement.
Proofreading:
An overview of the proofreading process is included in the online supplement.
Cultural Adaptation of the Images
In addition to the creation of culturally appropriate wording, 7 sets of images were produced for different global regions, including Mexico, the United States (Spanish-speaking population), the Philippines, Central and South America, Western Europe and Australia, India and Asia–Pacific, the Middle East, and Turkey. These images were tested in the same participants who were involved in the linguistic validation of the CERT, to determine if the participants thought that the images represented them. Prior to interviewing the participants, the questions were developed using a single forward translation of the questions from English into the target language. Participants were asked the following questions:
- Do you feel these images (skin color, eye color, hair color, hair texture, hair length, hair style, facial features, clothing, accessories, apparent age, etc.) represent you?
- Do you feel these images (skin color, eye color, hair color, hair texture, hair length, hair style, facial features, clothing, accessories, apparent age, etc.) represent patients with your condition?
- How do these images make you feel?
- Anything else you would like to share about these images?
The images were then updated based on the feedback according to different cultures, demographics, and countries via an iterative process.
Ethics Approval and Informed Consent
As this study exclusively involved translations, no personally identifiable information or protected health information was collected, and there was no drug intervention. Therefore, institutional review board approval was not needed. Additionally, study results were in tabular form, and aggregated analyses omitted participant identification. Prior to involvement in the linguistic and cultural validation of the CERT, participants were given a standardized form in their native language that explained the nature of the linguistic validation, how the cognitive debriefing interviews would be conducted, and how their feedback would be used. Details concerning privacy and data protection were also included. Informed consent was obtained from all participants who contributed to the linguistic and cultural validation of CERT translations. This study complied with all applicable laws regarding participant privacy, as described in the Declaration of Helsinki.
Results
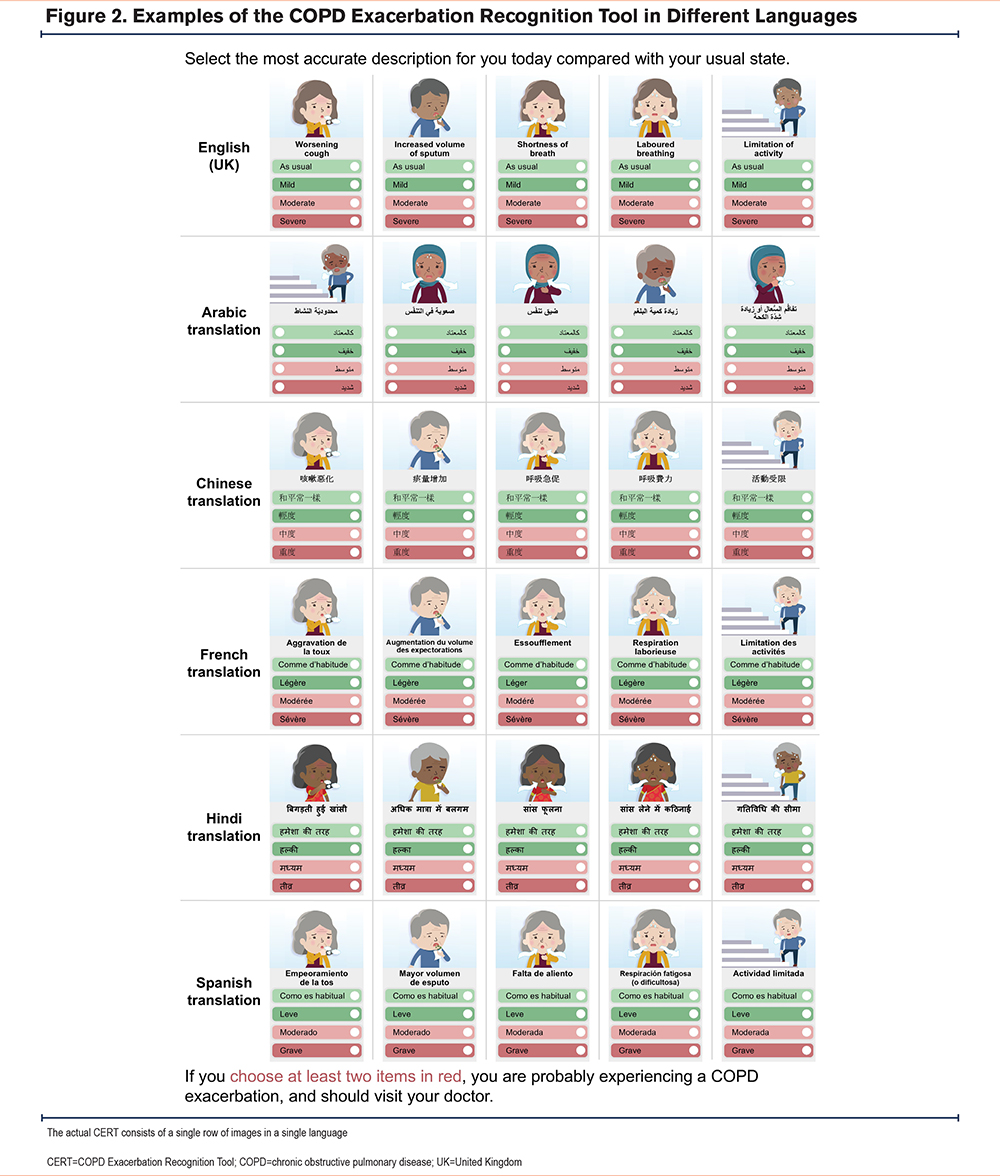
Examples of the CERT translated into different languages, along with representative images, are shown in Figure 2.
A total of 212 participants with COPD were involved in the whole translation program across all 46 CERT translations. An overview of the demographics and characteristics of participants included in the cognitive debriefing interviews for 5 CERT translations are included in Supplemental Table 2 in the online supplement.
During the cognitive debriefing stage (when participants were asked to reword the instructions, items, and responses in the CERT to confirm their understanding), most participants demonstrated good comprehension of the tool (Table 2; Supplemental Table 3 in the online supplement). In instances where phrases in a CERT translation were interpreted slightly differently than intended by a majority of participants, the wording was updated and retested among the participants. For example, in the Norwegian translation, most participants interpreted the translation of “shortness of breath” as “kortpustethet” to mean “difficulty breathing.” However, when this was updated to “åndenød,” participants interpreted this as feeling like there is not enough air, which was closer to the original meaning. The final wording was subsequently updated to “åndenød.”
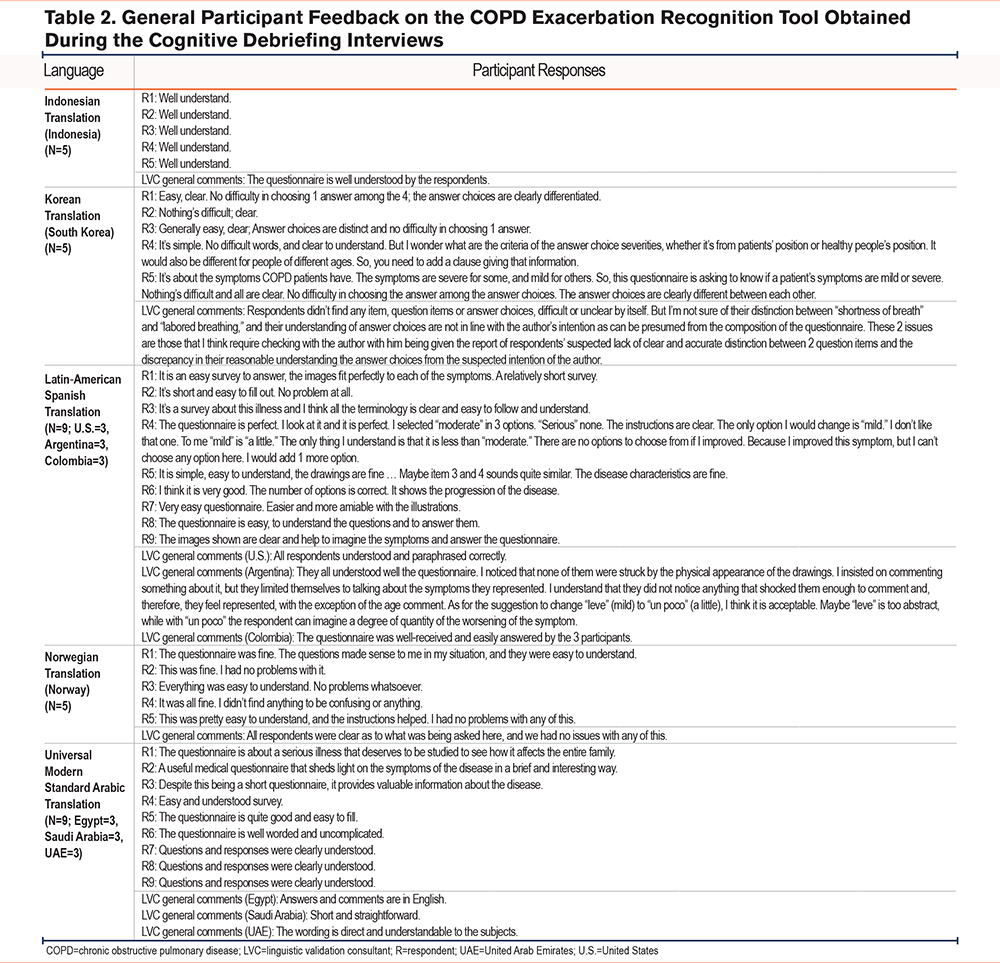
Overall, the CERT was well received by participants, and the translations were judged to be conceptually equivalent to the English source CERT and harmonized with each other (Table 2; Supplemental Table 3 in the online supplement). Most participants found the tool easy to use, with 54.5% of participants specifically describing the tool as clear, simple, and/or easy, and an additional 24.2% of participants commenting that the tool was well understood (Table 2).
Of the 30 participants who provided responses to all questions in the cognitive debriefing interviews of the Universal Arabic, Korean, Indonesian, Norwegian, and Latin-American Spanish translations of the CERT, 24 (80.0%) agreed that the images were representative of themselves (Table 3). In particular, participants commented that the presentation of the symptoms in the CERT corresponded to how they and/or other people with COPD usually appear when experiencing the same symptoms. A total of 4 (13.3%) participants who did not find the images to be representative of themselves responded that the images were likely to represent other patients with COPD instead. However, 4 participants (13.3%) commented that people in the images looked younger than themselves and/or other people with COPD.

Discussion
COPD exacerbations are often under-reported by patients.6,11 Possible reasons may include lack of patient awareness early in their diagnosis of what an exacerbation is, and inadequate provision of information on how to identify exacerbations (Supplemental Tables 5A and 5E in the online supplement). Therefore, access to tools like the CERT is important if patients are to better recognize the early signs of an exacerbation and seek prompt assessment and treatment. This study linguistically and cross-culturally validated 46 CERT translations across 25 countries and 6 continents and found that they appeared to be conceptually equivalent to the original CERT and harmonized with each other. Participants found the CERT easy to use and understand and confirmed that the images were representative of themselves and/or other people living with COPD. This was the largest of several translation projects for the CERT to date and brings the total number of translations to over 60, which are all now available on the Global Allergy and Airways Patient Platform (GAAPP) website22 (Supplemental Table 4 in the online supplement).
Images included alongside the text in the CERT can aid patient understanding of medical terminology (Supplemental Table 5C in the online supplement), and the addition of pictures in health care communications has been shown to improve comprehension and adherence to health instructions.23 The images were culturally adapted according to participant input to ensure that they reflected patients from a diversity of cultures, countries, and religions, aiming to improve global accessibility and health literacy. The majority of participants specifically commented that the CERT is easy, clear, and/or simple, or commented that the tool was well understood. The tool may be particularly useful in helping newly diagnosed people with COPD or people who are unable to access care easily (Supplemental Tables 5B and 5D in the online supplement). However, 13.3% of participants who provided responses in the cognitive debriefing interviews regarding the Universal Arabic, Korean, Indonesian, Norwegian, and Latin-American Spanish translations of the CERT found that the images of the people in the tool appeared younger than themselves and/or people with COPD. This may be due to the cartoon nature of the images. As the majority of participants found the images representative of themselves and/or other participants with COPD, no changes were made.
Although subtleties had to be captured when translating the CERT, the tool contains only a small number of symptoms that are universally experienced by patients with COPD. Participants who reviewed the CERT during the cognitive debriefing stage would have been able to recognize the symptoms that they experience during a COPD exacerbation and to think of appropriate words that they themselves would use. This is particularly important as a previous study identified a disconnection between patients’ experiences of exacerbations and the language and definitions for exacerbations used in clinical practice.6
While the CERT is now accessible to patients in over 60 languages, it is necessary to continue the process of translation and validation into as many languages and regional dialects as possible. The alternative of producing a CERT in every country would require extensive research and the close similarity between the original CERT developed in China and the CERT-J developed in Japan suggests that similar items would be identified in other countries.13,15 We argue that the process of translating and validating the tool, as described here, is a proven methodology that is in accordance with FDA guidance and industry standards.17-19 Furthermore, using translated versions derived from a single original version ensures a consistent approach across countries. In this context, it should be noted that the Predictors of Acute COPD Exacerbation (PACE) study, which demonstrated that the tool can reliably identify the onset of an acute exacerbation of COPD one day before clinical diagnosis with good sensitivity and specificity, was performed using a German translation of the CERT.16
One potential limitation of the translation and cultural and linguistic validation of the CERT is that the participants included in the cognitive debriefing stage may have had a respiratory disease other than COPD; therefore, in the cultural and linguistic validation of the Korean CERT translation, the terms “chronic bronchitis” and “emphysema” were used. However, the results of the cultural and linguistic validation of the Korean CERT translation were consistent with other translations, so we do not think that this will have affected the results. Another potential limitation of the study is that when participants were interviewed to determine if the images included in the tool had been sufficiently culturally adapted, they were not asked whether the images also aided their understanding of the tool. Future translation and linguistic validation studies of patient education tools should consider including a question in the participant interviews to determine if imagery improves participants' comprehension of the tool as previous research suggests.2,3
While there is scope to expand the translation and linguistic validation of the CERT by testing the items included in the tool in each individual country, the results of our study and the recent PACE study16 indicate that this is unnecessary. Additional languages for CERT translation continue to be considered and country-specific implementation programs are in progress; it is anticipated that these translations and programs will further facilitate patient accessibility to the tool.
Conclusions
This study cross-culturally and linguistically validated 46 CERT translations across 25 countries and 6 continents. The CERT is easy for patients to understand, and the translations can be considered to be equivalent regardless of country or language. These translations may support health care professionals in multiple global regions in educating patients to recognize and act upon early signs of a COPD exacerbation.
Acknowledgements
Authors’ contributions: MM, TM, PJ, RG, MT, and MH were involved in the study conception or design, acquisition of data, and data analysis or interpretation. RT-S was involved in the data interpretation and in creating the GAAPP Governance Board. PD and RW provided testimonials of their experiences living with COPD. All authors were involved in reviewing and editing the manuscript prior to submission, significantly contributed to the intellectual content of the article, and approved the final version to be published.
Data Availability: For requests for access to anonymized patient level data, please contact the corresponding author. No other additional documents, such as the study protocols, will be made available.
Other acknowledgements: The authors would like to thank Alfons Viñuela for his insights into this study. Editorial support (in the form of writing assistance, including preparation of the draft manuscript under the direction and guidance of the authors, collating and incorporating authors’ comments for each draft, assembling tables and figures, grammatical editing, and referencing) was provided by Sarah Case of Luna, OPEN Health Communications, and was funded by GSK in accordance with Good Publication Practice guidelines. CERT copyright information: 2023 the GSK group of companies or its licensor.
Declaration of Interest
Rainer Gloeckl is a member of the CERT governance board which is led by GAAPP. Ruth Tal-Singer is a retiree and shareholder of GSK and reports share options from ENA Respiratory, and personal fees from AstraZeneca, Boehringer Ingelheim, the COPD Foundation, ENA Respiratory, GAAPP, Global Skin, GSK, ImmunoMet, ItalyAndBeyond, Janssen, Renovion, Roche, Samay Health, Teva, and Vocalis Health. Peter Deussen has received fees from Biomedical Research Group Oxford and GAAPP for public and patient involvement. Russell Winwood is a patient consultant for Inogen Global. Tharishini Mohan, Megan Turner, Mohamed Hamouda, and Mandeep Moore are employees of and/or hold financial equities in GSK. Paul Jones is a contractor to and holds financial equities in GSK.