Running Head: Bleeding Risks With Warfarin Versus DOACs in COPD-AF
Funding Support: Fund for Health Research of Fujian Province (Grant number: 2023-63)
Date of Acceptance: February 9, 2026 | Published Online Date: February 23, 2026
Abbreviations: AECOPD=acute exacerbation of chronic obstructive pulmonary disease; AF=atrial fibrillation; ALP=alkaline phosphatase; ALT=alanine aminotransferase; aOR=adjusted odds ratio; AR=absolute risk; ARB=angiotensin 2 receptor blocker; AST=aspartate aminotransferase; ATO=average treatment effect in the overlap population; BMI=body mass index; CCB=calcium channel blockers; CHD=coronary heart disease; CI=confidence interval; COPD=chronic obstructive pulmonary disease; DOACs=direct oral anticoagulants; eGFR=estimated glomerular filtration rate; EMR=electronic medical record; FXa=factor Xa; Hb=hemoglobin; HF=heart failure; IPTW=inverse probability of treatment weighting; IQR=interquartile range; ISTH=International Society on Thrombosis and Haemostasis; OR=odds ratio; NACE=net adverse clinical events; NNH=number needed to harm; NNT=number needed to treat; NSAIDs=nonsteroidal anti-inflammatory drugs; NVAF=nonvalvular atrial fibrillation; OACs=oral anticoagulants; OW=overlap weighting; PE=pulmonary embolism; PLT=platelet count; PPI=proton pump inhibitor; PS=propensity score; PSM=propensity score matching; RD= risk difference; SE=systemic embolism; SMD=standardized mean difference; TBil=total bilirubin; TIA=transient ischemic attack; VTE=venous thromboembolis
Citation: Zhao N, Wei T, Huang X, et al. Comparison of bleeding risks and all-cause death between warfarin and direct oral anticoagulants in patients with atrial fibrillation and chronic obstructive pulmonary disease: a multicenter retrospective cohort study. Chronic Obstr Pulm Dis. 2026; 13(2): 93-103. doi: http://doi.org/10.15326/jcopdf.2025.0648
Online Supplemental Material: Read Online Supplemental Material (763KB)
Introduction
Atrial fibrillation (AF) is the most common sustained cardiac arrhythmia, and its incidence and prevalence have been increasing globally in recent years.1 Ischemic stroke and thromboembolism in other parts of the body are the main causes of morbidity and mortality in patients with AF,2 with AF increasing the risk of ischemic stroke by 3 to 5 times. It is estimated that 15% of strokes worldwide are attributable to AF.3 To prevent thrombotic events such as ischemic stroke, patients with AF typically receive anticoagulant medications for prophylactic treatment.1,4 However, anticoagulation inevitably carries the risk of bleeding, with severe bleeding potentially being fatal.5,6 The incidence of AF increases with age, and therefore, patients often have multiple comorbidities. Apart from cardiovascular and cerebrovascular diseases, chronic obstructive pulmonary disease (COPD) is a common comorbidity in patients with AF, affecting about 13% of these patients.7,8
COPD is a heterogeneous lung disease caused by smoking and the inhalation of toxic particles and gases indoors and outdoors, leading to persistent airflow obstruction and respiratory symptoms.9 It is characterized by localized inflammation in the lungs to inhaled substances (such as cigarette smoke and air pollutants) and even systemic inflammation.10 Inflammatory diseases, as risk factors for venous thromboembolism (VTE), can induce thrombosis.11 VTE is the most common and potentially fatal complication in COPD patients hospitalized for exacerbation.12 Apart from VTE, the incidence of pulmonary embolisms (PEs) in COPD patients should not be overlooked. A population-based cohort study showed that the incidence of PEs in COPD patients is 12.31/10,000 person-years, which is approximately 4 times higher than in those without COPD.13 In patients with acute exacerbations of chronic obstructive pulmonary disease (AECOPDs), there is a significant correlation between blood coagulation and anticoagulation processes as well as between oxidative and antioxidative mechanisms. Early anticoagulation may improve the prognosis of these patients. Using low molecular weight heparin or oral anticoagulation may improve lung function and slow disease progression in patients with AECOPDs.14 Current guidelines and the Global initiative for chronic Obstructive Lung Disease (GOLD)9 support anticoagulation therapy for patients with AECOPDs. However, to date, there is no consensus on prophylactic anticoagulation therapy in COPD patients.9
Due to the high risk of ischemic stroke and other thromboembolic events in patients with AF, those with concurrent COPD must undergo prophylactic anticoagulation therapy. However, COPD is not merely a localized inflammatory disease of the lungs but a systemic inflammatory condition. Owing to persistent airway obstruction and decline in lung function, such patients often experience prolonged hypoxemia, chronic inflammatory responses,9,15 and long-term use of inhaled medications and other treatments. These factors may alter coagulation mechanisms, thereby increasing the risk of bleeding. Pulmonary diseases may lead to hemoptysis or bronchial bleeding, and anticoagulation could exacerbate these conditions, increasing the risk of severe bleeding events. Studies have found that AF patients with COPD have a higher risk of major bleeding compared to those without COPD.16 Different anticoagulants, especially warfarin and direct oral anticoagulants (DOACs), vary in their mechanisms of action and metabolic pathways, thus differing in bleeding risks. Therefore, the choice of oral anticoagulants is particularly crucial for AF patients requiring prophylactic treatment when combined with COPD.
In recent years, studies exploring the efficacy and safety of different types of oral anticoagulants in patients with AF and other cardiovascular diseases (such as hypertension, chronic coronary artery disease), chronic kidney disease, and liver dysfunction have been published.17-20 However, there is a lack of research focused on AF patients with COPD. The risks of bleeding and all-cause death associated with different oral anticoagulants in patients with AF and COPD remain unclear. Therefore, based on real-world data, we conducted a multicenter retrospective cohort study aimed at comparing the risks of bleeding and all-cause death (including major bleeding, minor bleeding, total bleeding, and all-cause death events) between warfarin and DOACs in patients with AF and COPD undergoing anticoagulation.
Methods
Study Design
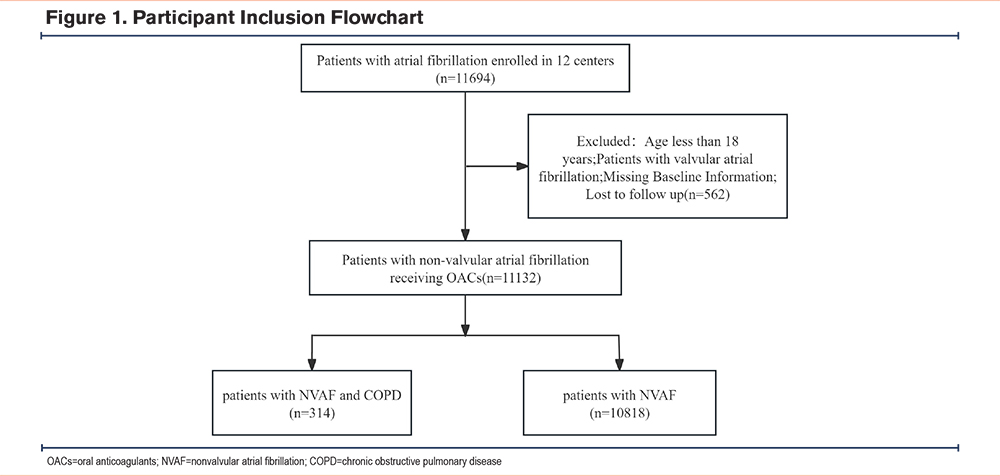
From January 2016 to December 2020, we conducted a retrospective cohort study across 12 centers in China to analyze the risks of bleeding and all-cause death in patients with AF and COPD who were treated with different oral anticoagulants, the list of multicenter hospitals is provided in Supplementary Table 1 in the online supplement. The institutional review board of Fujian Maternal and Child Health Hospital approved the protocol (Registration No.: ChiCTR 2300067734). As the study was retrospective, the institutional review board waived the requirement for patient informed consent. The inclusion criteria for the study were patients diagnosed with AF, and patients currently receiving treatment with warfarin or DOACs (including dabigatran, rivaroxaban, apixaban, and edoxaban). The exclusion criteria were: (1) patients with valvular AF, (2) patients aged ≤18 years, (3) missing baseline information, and (4) loss to follow-up. Ultimately, 11,132 patients receiving oral anticoagulant therapy for AF were included in this study, with an average follow-up period of about 13 months. The flowchart of participant inclusion is shown in Figure 1. These patients were divided into 2 subgroups: patients with AF and concomitant COPD (n=314) and patients with AF without COPD (n=10,818).
Data Collection and Definition
Researchers at each center involved in this study retrieved patients with AF through the hospital medical information system and conducted follow-ups with patients or their families to ascertain clinical outcomes. The data collected included demographic information (such as age, gender, body mass index [BMI], smoking, alcohol consumption, etc.), biochemical indicators (such as total bilirubin, alanine transaminase, aspartate transaminase, albumin, etc.), information on comorbidities (such as hypertension, diabetes, coronary heart disease, vascular diseases, etc.), and information on concomitant medications (such as statins, antiplatelet drugs, proton pump inhibitors [PPIs], etc.). Before follow-up, we standardized the follow-up questions and strictly defined each clinical event. The follow-up period was defined as from the start date of oral anticoagulant therapy to the stop date of oral anticoagulant therapy or the end date of the study. Identification and diagnosis of COPD were primarily based on physician-established diagnoses recorded in the hospital electronic medical record (EMR) system. Bleeding events were ascertained from the hospital EMR during inpatient or outpatient encounters, and telephone follow-up after discharge. We prioritized objective chart documentation when ascertaining bleeding; for out-of-hospital bleeding reported during follow-up, we sought to obtain supporting medical records for verification whenever feasible. Additionally, based on the clinical data of the patients, we assessed the patients' CHA2DS2-VASC score21 which includes congestive heart failure/left ventricular dysfunction, hypertension, age ≥75 years, diabetes, stroke or transient ischemic attack, vascular disease, age 65–74 years, and female gender, and their HAS-BLED score which includes hypertension, abnormal liver function, abnormal renal function, stroke, bleeding, age >65 years, and drugs and alcohol. The primary exposures were presence of COPD and oral anticoagulant class, warfarin versus DOACs; DOACs were further categorized as factor XA (FXa) inhibitors (rivaroxaban, apixaban, edoxaban) and dabigatran.
Study Outcomes
The main outcome events of this study were total bleeding, major bleeding, minor bleeding, all-cause death events, and net adverse clinical events (NACE), defined as a composite endpoint of thromboembolic events or major bleeding (counted as positive if either component occurred). According to the definition by the International Society on Thrombosis and Haemostasis (ISTH), major bleeding refers to bleeding that leads to death, occurs in a critical organ (intracranial, intraspinal, intraocular, retroperitoneal, intra-articular, pericardial, or involves compartment syndrome), or results in a decrease in hemoglobin levels of at least 2g/dl or the transfusion of at least 2 units of whole blood or red cells.22 Minor bleeding was defined as any overt bleeding that did not meet the ISTH criteria for major bleeding and did not fulfill the ISTH criteria for clinically relevant nonmajor bleeding.23 Thromboembolic events included ischemic stroke and systemic embolism (SE). SE was defined as acute vascular occlusion of an extremity or organ confirmed by imaging, surgery, or autopsy.24 Total bleeding includes all bleeding events (major and minor). All suspected bleeding and thromboembolic events were classified according to ISTH criteria, with objective documentation in the medical record serving as the primary source of confirmation. For out-of-hospital events identified solely via telephone follow-up, we sought to retrieve medical evidence for verification whenever feasible. All study endpoints were assessed for occurrence over the entire follow-up period. Bleeding and NACE were treated as binary outcomes. Owing to the lack of precise event-time data, all-cause death was likewise analyzed as a binary outcome, defined as any death occurring from cohort entry through the end of follow-up.
Statistical Analysis
Continuous variables were assessed for normality. Normally distributed data are presented as mean ± standard deviation and compared using Student’s t test; non-normally distributed data are summarized as median (interquartile range) and compared using the Mann-Whitney U test. Categorical variables are presented as counts and percentages and compared using the χ2 test. To evaluate the impact of COPD on bleeding, all-cause mortality, and NACE, we analyzed the overall AF cohort (n=11,132) using COPD status as the exposure and applied propensity score overlap weighting (OW) with overlap-weighted logistic regression as the primary approach. As sensitivity analyses, we performed 1:1 propensity score matching (PSM) and re-estimated OR/absolute risk (AR)/risk difference (RD) to assess consistency across causal inference strategies.
In the AF with comorbid COPD subcohort (n=314), with exposure defined as DOACs versus warfarin, the primary analysis likewise used OW with overlap-weighted logistic regression. Covariate balance was assessed using the absolute standardized mean difference (SMD) with a threshold of 0.10; any covariate with residual imbalance (|SMD|>0.10) was included in the weighted outcome model. We reported adjusted odds ratios (aORs) and ARs/RDs with 95% confidence intervals (CIs) obtained via marginal standardization (g-computation). Sensitivity analyses comprised: (1) an unweighted multivariable logistic regression, (2) logistic regression with inverse probability of treatment weighting (IPTW), and (3) a restriction excluding baseline concomitant antiplatelet therapy. To address rare events or separation, we used cluster-robust standard errors by center and, when necessary, applied Firth-penalized logistic regression and mixed-effects logistic regression (center as a random intercept) as fallback models.
Within the AF with comorbid COPD cohort, using OW we further evaluated the effectiveness and safety stratified by renal function (DOAC versus warfarin), and, among patients treated with DOAC, an exploratory comparison of FXa inhibitors versus dabigatran. Each subgroup reported aOR and AR/RD (95% CI). When the RD and its 95% CI were directionally concordant and clinically interpretable, we calculated the numbers needed to treat and number needed to harm (NNH). All statistical analyses were conducted in R (version 4.4.3).
Results
Baseline Characteristics
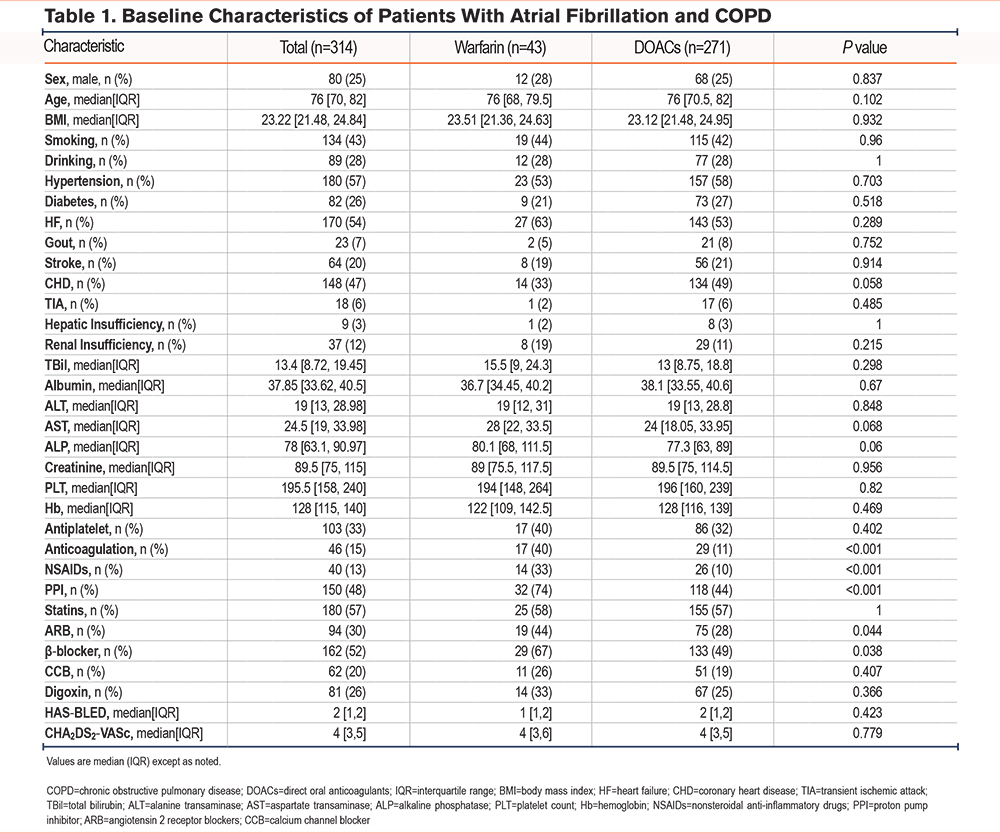
The baseline characteristics of AF patients on anticoagulant therapy, with and without COPD, are shown in Supplementary Table 2 in the online supplement. Compared with the AF-only group (n=10,818), the AF with comorbid COPD group (n=314) was older, had a higher proportion of men, and a lower BMI; it also carried a greater comorbidity burden, with heart failure, coronary artery disease, and prior stroke being more common. Overall use of oral anticoagulation was significantly lower. In laboratory indices, total bilirubin, alanine aminotransferase, and aspartate aminotransferase were lower, whereas serum creatinine was higher; platelet counts did not differ significantly. Regarding medications, use of digoxin and nonsteroidal anti-inflammatory drugs (NSAIDs) was more frequent, whereas β-blockers and calcium channel blockers were used less often; the use of antiplatelet agents, statins, and angiotensin 2 receptor blockers (ARBs) was similar. Risk scores (HAS-BLED and CHA2DS2-VASc) were higher. Except for smoking status and platelet count, most between-group differences were statistically significant (p<0.05). Within the COPD group, baseline characteristics between patients treated with warfarin (43 patients) and direct oral anticoagulant (271 patients) were compared among a total of 314 patients (Table 1). No differences were found between the 2 subgroups in terms of gender, age, BMI, smoking, other comorbidities, laboratory indicators, bleeding risk (HAS-BLED score), and stroke risk (CHA2DS2-VASc score). In terms of concomitant medication, compared to the warfarin group, patients on DOACs were more likely to also receive anticoagulants, NSAIDs, PPIs, ARBs, and β-blockers, with statistically significant differences (p <0.05). Event counts for the full cohort, stratified by anticoagulant, are shown in Supplementary Table 3 in the online supplement.
Impact of COPD on Clinical Outcomes in Patients With Atrial Fibrillation
In the overlap-weighted primary analysis stratified by anticoagulation strategy (warfarin versus DOACs), COPD was associated with a higher bleeding risk among patients treated with warfarin: total bleeding OR 2.53 (95% CI, 1.00–6.45), corresponding RD 9.05% (95% CI, 0.15%–22.50%); minor bleeding OR 3.00 (1.09–8.24), RD 8.53% (0.56%–21.53%). In the DOAC group, these associations were not significant. No significant associations were observed for major bleeding, all-cause death, or NACE in either anticoagulation stratum. The treatment-by-disease interaction was borderline and not statistically significant for total bleeding (P=0.057) and minor bleeding (P=0.061) (Figure 2). Overlap-weighted event counts and absolute risks are shown in Supplementary Table 4 in the online supplement.
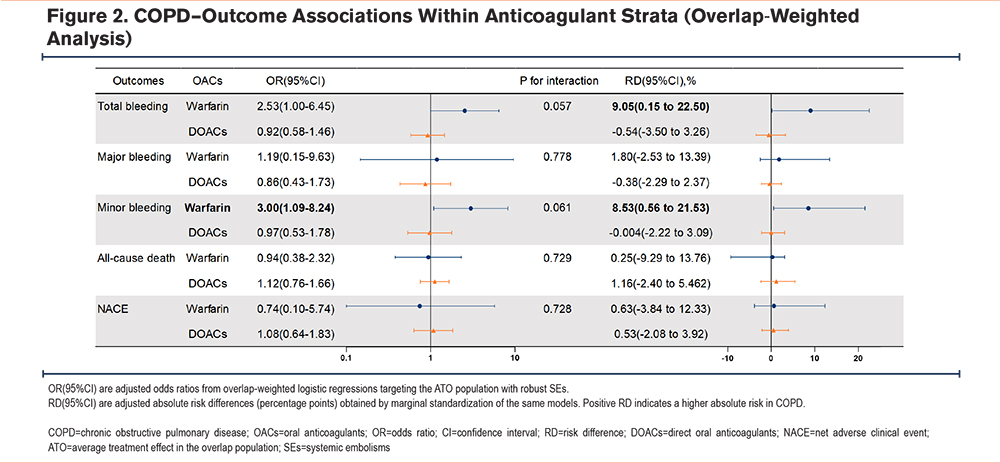
Covariate balance assessment indicated that OW reduced all covariate |SMD| values to <0.10; PSM (1:1) generally met the threshold, although several variables showed residual imbalance. Baseline differences between matched-included and matched-excluded patients are provided in Supplementary Table 5 in the online supplement. In the unweighted sample, there were pronounced imbalances in age, anticoagulant type, creatinine, HAS-BLED score, heart failure, coronary artery disease, BMI, and albumin (Supplementary Figure 1 in the online supplement). Accordingly, OW was selected as the primary analysis to enhance the robustness of the effect estimates.
As sensitivity analyses, the crude model and PSM (1:1) yielded results consistent with the primary findings: among patients treated with warfarin, COPD was associated with higher risks of overall and minor bleeding, whereas no such associations were observed among DOAC; and there were no consistent significant associations with major bleeding, all-cause death, or NACE. The interaction for overall and minor bleeding was borderline in these sensitivity analyses as well, with directions concordant with the primary analysis (Supplementary Table 6 in the online supplement).
Impact of Oral Anticoagulants on Clinical Outcomes in Patients With Atrial Fibrillation and COPD
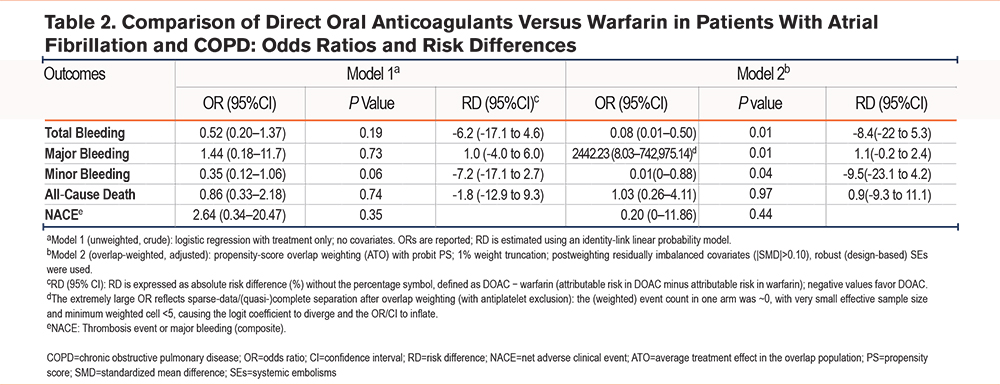
In patients with AF and COPD, the OW-adjusted primary analysis (Model 2) showed that, compared with warfarin, DOACs were associated with a lower risk of total bleeding (OR, 0.08; 95% CI, 0.01–0.50; RD, -8.4%; 95% CI, -22.0% to -5.3%). Minor bleeding was likewise reduced (OR 0.01; RD, -9.5%; 95% CI, -23.1% to -4.5%), although the CIs were wide. Estimates for major bleeding were unstable because of sparse data (near-zero weighted events in the treatment arm); the overlap-weighted event counts are provided in Supplementary Table 7 in the online supplement. The RD for major bleeding was close to zero, with CIs crossing the null. No clear differences were observed for all-cause death or NACE.
Model 1 (crude; unweighted and unadjusted) showed no statistically significant differences for any endpoint, but the trends for overall and minor bleeding (OR <1) were consistent with the primary analysis (Table 2). As sensitivity analyses, Model 3 (unweighted but covariate-adjusted) demonstrated significant advantages of DOACs in reducing total bleeding (OR, 0.27) and minor bleeding (OR, 0.18), further supporting the primary findings. Model 4 (IPTW) yielded results concordant with the primary analysis, again confirming the advantages of DOACs for overall and minor bleeding (OR, 0.21 and 0.09, respectively). Model 5 (OW excluding patients on baseline antiplatelet therapy) was markedly affected by sparse data, rendering several outcomes (e.g., major bleeding) nonestimable or yielding aberrant ORs, reflecting reduced statistical power after the restriction; nonetheless, the RDs were directionally and quantitatively similar to the other models. Across methods, all-cause death and NACE showed no stable differences (effects near zero with wide CIs), as detailed in Supplementary Table 8 in the online supplement. Balance diagnostics (Supplementary Figure 2 in the online supplement) indicated that OW achieved |SMD| ≤0.10 for all covariates, IPTW performed second best, and substantial imbalances persisted in the unweighted sample.
Subgroup Analyses
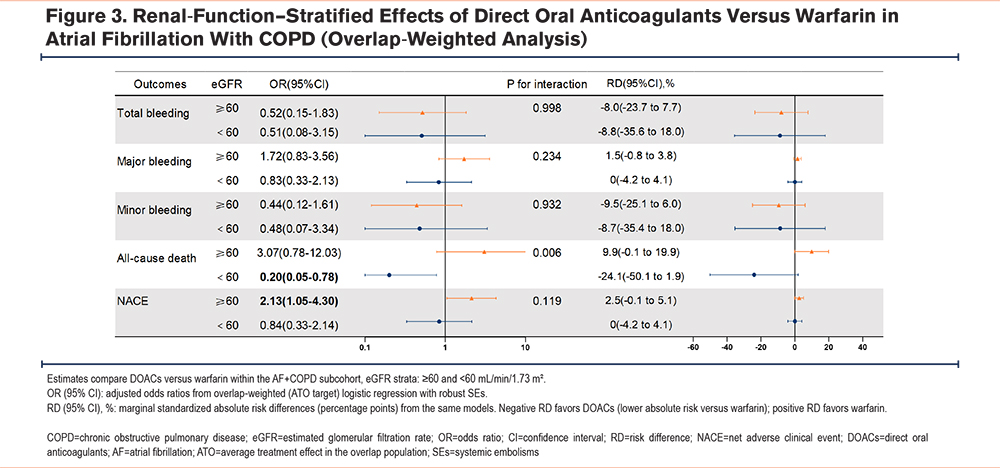
Based on the propensity score overlap-weighted model, a subgroup analysis stratified by baseline renal function (estimated glomerular filtration rate [eGFR]) demonstrated a significant interaction for all-cause death (P for interaction=0.006). Among patients with eGFR ≥60mL/min/1.73m2, DOACs were associated with a higher risk of all-cause death compared with warfarin (OR, 3.07; 95% CI, 0.78–12.03; RD, 9.9%). In contrast, among those with eGFR <60mL/min/1.73m2, DOACs were associated with a significantly lower mortality risk (OR, 0.20; 95% CI, 0.05–0.78; RD, -24.1%). For overall and minor bleeding, DOACs showed lower risks in both strata with negative RDs, and interaction P-values >0.05. Major bleeding did not differ significantly between DOACs and warfarin in either stratum. The direction of the composite endpoint NACE paralleled that of all-cause death, although the interaction was not significant (P=0.119). Detailed ORs and RDs are presented in Figure 3, and overlap-weighted event counts and ARs are summarized in Supplementary Table 9 in the online supplement.
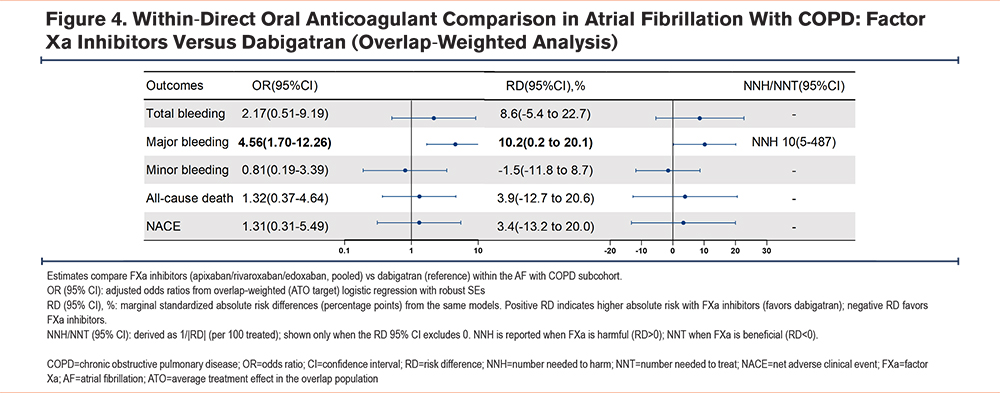
In the AF with comorbid COPD population restricted to DOAC, an overlap-weighted model with adjustment for residually imbalanced covariates compared FXa inhibitors (apixaban/rivaroxaban/edoxaban) versus dabigatran. Major bleeding was significantly higher with FXa inhibitors (OR, 4.56; 95% CI, 1.70–12.26; RD, 10.2%; 95% CI, 0.2%–20.1%), corresponding to an NNH of 10 (95% CI, 5–487). Total bleeding trended higher but was not significant (OR, 2.17; 95% CI, 0.51–9.19; RD, 8.6%; 95% CI, -5.4% to 22.7%). Minor bleeding, all-cause death, and NACE did not differ significantly (ORs 0.81, 1.32, and 1.31, respectively; corresponding RDs -1.5%, +3.9%, and +3.4%, with 95% CIs spanning the null). Detailed ORs and RDs are presented in Figure 4, and overlap-weighted event counts and ARs are shown in Supplementary Table 10 in the online supplement.
Discussion
This study is a multicenter retrospective cohort study based on 12 hospitals in China, aimed at comparing the risks of bleeding and all-cause death between warfarin and DOACs in patients with AF and COPD undergoing anticoagulation therapy. We used propensity score overlap-weighted (average treatment effect in the overlap population [ATO]) logistic regression as the primary analytic framework, reported ARs and RDs with 95% CI and accordingly derived the NNH. Sensitivity analyses were performed to enhance the robustness of inference.
In overlap-weighted, anticoagulant-stratified analyses (with concordant sensitivity checks), COPD was linked to higher bleeding risk only among warfarin; major bleeding, all-cause death, and NACE showed no consistent associations, and the treatment-by-disease interaction for bleeding was borderline. Within AF and COPD, DOACs versus warfarin reduced overall and minor bleeding, with no stable differences for major bleeding, all-cause death, or NACE (major bleeding estimates were unstable due to rarity).
Renal function modified the death effect: DOACs lowered mortality for eGFR <60mL/min/1.73m2, with no advantage at eGFR≥60mL/min/1.73m2. Among DOAC, FXa inhibitors versus dabigatran increased major bleeding, while other endpoints were similar.
In line with our findings, Romiti et al7 and others have found that patients with COPD have higher risks of all-cause death and bleeding. A 2018 study16 indicated that COPD adversely affects the prognosis of AF patients, increasing the risks of all-cause death, cardiovascular disease hospitalization, and cardiovascular death. COPD patients often have additional risk factors such as smoking, old age, comorbidities, and concomitant medications that may increase bleeding risks. Smoking is not only a key risk factor for COPD but also shows causal and dose-response relationships with the initiation, perpetuation, and recurrence of AF. A recent study25 reported that for every additional 10 cigarettes smoked per day, AF risk increases by approximately 14%, suggesting that smoking promotes arrhythmogenesis and thrombogenicity via inflammatory mechanisms, atrial structural remodeling, and electrophysiological pathways. The impact of smoking on warfarin is relatively well established: polycyclic aromatic hydrocarbons in tobacco smoke induce CYP1A2 and other enzymes, accelerating metabolism (principally of the R-warfarin enantiomer), thereby increasing maintenance dose requirements and attenuating anticoagulant effect.26 A meta-analysis estimated that smokers require, on average, a 12% higher warfarin maintenance dose; moreover, the international normalized ratio should be closely monitored and doses adjusted when patients quit smoking or change smoking intensity—an issue of particular relevance in COPD populations with fluctuating tobacco exposure.27 For DOACs, direct evidence that smoking per se alters drug exposure is limited. However, during acute COPD exacerbations, patients frequently receive macrolide or fluoroquinolone antibiotics and other agents, some of which are strong or moderate CYP3A4 inhibitors or inducers. These drug–drug interactions can substantially modify exposure to apixaban and rivaroxaban and increase bleeding risk (e.g., higher bleeding risk with concomitant clarithromycin). Thus, in real-world care of smokers with COPD, indirect interaction pathways warrant heightened vigilance.28
Since the introduction of DOACs, many clinical studies and meta-analyses have shown that the efficacy and safety of DOACs are comparable or even superior to warfarin in patients with AF, including those with additional comorbidities.29,30 Although current guidelines and the GOLD support anticoagulation therapy for patients with an AECOPD, a consensus on prophylactic anticoagulation therapy for COPD patients has not yet been reached.9 From the standpoint of biological plausibility, some investigators have hypothesized that vitamin K antagonists may be linked to vascular calcification by impairing activation of extrahepatic vitamin K–dependent proteins such as matrix Gla protein, whereas DOACs do not act through this pathway.31-34 However, this study did not capture key information—warfarin time in therapeutic range, DOAC dose appropriateness, or vascular imaging/biomarker data—needed to evaluate that mechanism. Accordingly, these considerations are offered solely as hypothesis-generating background, not as causal explanations or as a basis for our conclusions, and they require prospective validation.
Our study drew on a multicenter, real-world cohort. We used propensity score overlap weighting to enhance comparability and reported marginal (g-computation) and absolute effects (ARs/RDs), alongside harmonized methodological sensitivity analyses and key subgroup analyses within a unified framework; directions of effect were concordant. Nevertheless, several limitations merit consideration. First, endpoint ascertainment relied on in-hospital electronic medical records and postdischarge telephone follow-up. We prioritized objective chart documentation and, whenever possible, retrieved emergency/outpatient records and laboratory reports for verification; discrepant cases were reviewed by a second investigator. However, recall bias may persist for out-of-hospital events lacking complete documentation, and adjudication was not fully blinded. Second, the database lacked key clinical variables, limiting confounding control and assessment of prescribing quality. Third, sparse events and diminished overlap in certain subgroups/outcomes yielded unstable estimates with wide confidence intervals; moreover, the absence of precise event-time data precluded time-to-event analyses, restricting us to odds ratios and absolute risks over the follow-up period. Finally, as an observational study, residual and unmeasured confounding cannot be excluded, and generalization should be made cautiously.
Conclusion
Among anticoagulated patients with AF and COPD, overlap-weighted (ATO) analyses showed that DOACs, compared with warfarin, were associated with lower risks of overall and minor bleeding, with no clear differences in major bleeding, all-cause mortality, or NACE. The effect on mortality differed by renal function: a benefit was observed at eGFR <60mL/min/1.73m2 but not at eGFR≥60mL/min/1.73m2. Within the DOAC class, FXa inhibitors were associated with a higher risk of major bleeding than dabigatran. These findings support preferential use of DOACs to minimize bleeding, suggest a possible survival benefit in renal impairment, and warrant prospective confirmation; agent selection should consider NNH and individual patient factors.
Acknowledgements
Author contributions: NZ and TW contributed equally to the conception and design of the study, data acquisition, analysis and interpretation, and drafting the manuscript. XH, GW, RL, QZ, XLiu, HD, XLin, YLiu, JS, XD, CY, SJ, and YLan contributed to data acquisition and critical revision of the manuscript. JZ contributed to the conception and design, supervision of the study, and critical revision of the manuscript. All authors approved the final version.
Data sharing statement: The data sets used and/or analyzed for the study described in this manuscript are available upon reasonable request. Qualified researchers may request access to patient level data and related study documents including the study protocol and the statistical analysis plan. Patient level data will be deidentified and study documents will be redacted to protect the privacy of trial participants and to protect commercially confidential information.
Declaration of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.