Running Head: Isoflavones and COPD
Funding Support: Research reported in this publication was supported by the National Institute on Minority Health and Health Disparities of the National Institutes of Health under award number P50MD010431. This article was developed under Assistance Agreement No.83615001 awarded by the U.S. Environmental Protection Agency to Nadia Hansel. The funding sources had no role in the study design, data collection, analysis, interpretation, or decision to submit the manuscript for publication.
Date of Acceptance: February 12, 2026 | Published Online Date: February 23, 2026
Abbreviations: 11dTxB2=11-dehydro-thromboxane B2; 8-OHdG=8-hydroxydeoxyguanosine; CAT=COPD Assessment Test; CCQ=Clinical COPD Questionnaire; CI=confidence interval; COPD=chronic obstructive pulmonary disease; CURE COPD=Comparing Urban and Rural Effects of Poverty on COPD; DHA=docosahexaenoic acid; ECSC=Ease of Cough and Sputum Clearance Questionnaire; ELISA=enzyme-linked immunosorbent assay; EPA=eicosapentaenoic acid; FEV1=forced expiratory volume in 1 second; FFQ=food frequency questionnaire; GEE=generalized estimating equation; IFN=interferon; IL=interleukin; IQR=interquartile range; IRR=incidence rate ratio; MCID=minimal clinically important difference; mMRC=modified Medical Research Council; SD=standard deviation; SGRQ=St George’s Respiratory Questionnaire; %pred=percentage predicted; TBARS=thiobarbituric acid reactive substances; TNF=tumor necrosis factor
Citation: Belz DC, Quiroz E, Woo H, et al. Isoflavone intake is associated with decreased chronic obstructive pulmonary disease morbidity. Chronic Obstr Pulm Dis. 2026; 13(2): 125-135. doi: http://doi.org/10.15326/jcopdf.2025.0695
Online Supplemental Material: Read Online Supplemental Material (186KB)
Introduction
Diet and nutrition have increasingly been recognized as modifiable risk factors for chronic lung diseases such as chronic obstructive pulmonary disease (COPD). A “Western” dietary pattern, characterized by high intake of refined grains, red and processed meats, fried foods and sugar-sweetened beverages, has been associated with increased risk of COPD, increased respiratory symptoms, and lower lung function.1,2 Conversely, a Mediterranean or “Prudent” dietary pattern, characterized by high intake of vegetables, fruit, fish, poultry, and whole grains, was associated with a decreased risk of COPD. Specific dietary components that may explain the difference in lung health between these 2 patterns have also been investigated. As an example, increased omega-3 fatty acid intake has been associated with decreased COPD morbidity in terms of exacerbation frequency and symptom burden.3 Another potential contributor to this finding are isoflavones, phenolic compounds produced by legumes and found in foods such as tofu and soy milk. Isoflavone intake has been associated with decreased risk of other chronic diseases such as coronary heart disease, with one proposed mechanism being inhibition of inflammatory cytokine release which may similarly provide benefit in an inflammatory disease such as COPD. Additionally, isoflavones downregulate platelet activation, which is increased in COPD and has been associated with respiratory morbidity4; however, these relationships have not yet been studied.5,6
To fill this gap, we sought to evaluate the association of isoflavone intake, as measured by a food frequency questionnaire (FFQ), and COPD morbidity among individuals enrolled in the urban arm of the Comparing Urban and Rural Effects of Poverty on COPD (CURE COPD) study, a prospective cohort study assessing the impact of air pollution and diet on low-income COPD patients residing in Baltimore, Maryland.
Methods
Study Population and Study Design
Participants were longitudinally followed as part of an observational cohort of former smokers to study the impact of diet and indoor air pollution on COPD, with clinic visits at baseline, 3 months, and 6 months and with monthly telephone interviews for the 6-month study period and an additional 3 months thereafter. Eligible participants were aged 40 or older with physician-diagnosed moderate-to-severe COPD based on spirometry (postbronchodilator forced expiratory volume in 1 second [FEV1] to forced vital capacity <0.7 and FEV1 percentage predicted[%pred] <80%), a smoking history of >10 pack years. Participants also either resided in census tracts with a poverty rate above 10%, had only public health insurance, or had completed high school education or less. Additional details regarding catchment area and participant recruitment have been previously published.7 The study protocols were reviewed and approved by the Johns Hopkins School of Medicine Institutional Review Board, and written informed consent was obtained from all participants prior to any data collection.
Demographic and Socioeconomic Data
Demographic data, including self-reported race, household income, and educational attainment were obtained via questionnaire at baseline. Information on neighborhood characteristics were obtained via geocoded address at baseline. Neighborhood poverty rate represented the percentage of all families in a census-tract with annual income below the federal poverty rate.8
Dietary Assessment
Dietary intake was assessed by the Harvard FFQ,9 which was administered by trained study staff at 3 clinic visits (baseline, 3 months, and 6 months). The average consumption frequency of food items in the previous year was inquired with 9 options provided, ranging from “Never, or less than once per month” to “6+ per day.” Total isoflavone intake was obtained by summing intake of genistein, daidzein, glycitein, formononetin, and biochanin A. The validity and reproducibility of the FFQ,10 the food composition database used to calculate isoflavone values,11 and previous studies have demonstrated a moderately strong correlation between self-reported intake of isoflavone-rich foods and urinary isoflavone metabolite excretion.5,12
Respiratory Outcomes
Validated respiratory outcome measures were assessed by trained staff longitudinally at each clinic visit. Medication use was assessed by participant self-report via questionnaire. Health status was assessed with the COPD Assessment Test (CAT)13 and the Clinical COPD Questionnaire (CCQ),14 cough symptoms were assessed with the Ease of Cough and Sputum Clearance Questionnaire (ECSC),15 dyspnea was assessed with the modified Medical Research Council (mMRC) scale,16 and respiratory-specific quality of life was assessed with the St George’s Respiratory Questionnaire (SGRQ).17 CAT ranges were from 0–40, mMRC from 0–5, SGRQ from 0–100, CCQ from 0–6, and ECSC from 0–25, with higher scores indicating worse impact of COPD. In addition, exacerbation data was prospectively monitored over 9 months with monthly telephone interviews. Moderate exacerbations were defined as an unscheduled doctor’s visit, antibiotic or systemic steroid use, or treatment in an urgent care facility due to COPD- related illness. Severe exacerbations were defined as any COPD-related emergency department visit or hospitalization. Spirometry was performed according to the American Thoracic Society protocol (Koko spirometer; nSpire) at each study visit.
Biomarker Assessment
Serum, ethylenediaminetetraacetic acid plasma, and urine specimens were collected at each study visit. Urine and serum samples were processed within 2 hours of collection, stored at -80⁰C, and analyzed in a single batch at the Johns Hopkins Institute for Clinical and Translational Research Core Laboratory. Serum biomarkers were measured using a custom 7-plex Luminex assay and analyzed on the Luminex MAGPIX platform (Luminex; Austin, Texas) at the University of Maryland Cytokine Core Laboratory. This multiplex assay included the following cytokines and ranges of detection: interleukin (IL)-6 (1.3–960pg/mL), IL-8 (1.3–1000pg/mL), IL-1β (5.9–1436pg/mL), tumor necrosis factor (TNF)-α (2.3–553pg/mL), interferon (IFN)-γ (18–13300pg/mL).
Urine samples were analyzed using enzyme-linked immunosorbent assay (ELISA) at the Johns Hopkins Institute for Clinical and Translational Research Core Laboratory including urine 11-dehydro-thromboxane B2 (11dTxB2; Enzo Life Sciences; Lyon, France;pg/mL), urine thiobarbituric acid reactive substances (TBARS; BioAssay Systems, Hayward, California), urine 8-hydroxydeoxyguanosine (8-OHdG; Antibody online GMBH), and 8-isoprostane (Cayman Chemical; Ann Arbor, Michigan). Each urine assay was adjusted by dividing the measured value by urine creatinine quantified by ELISA (Quidel Corporation; San Diego, California). Samples that had undetectable levels of a certain biomarker were assigned the value of limit of detection/√2 and samples that exceeded the range of the standard curve were diluted to obtain a numeric value. The measured biomarkers were categorized as markers of inflammation (IL-6, IL-8, TNF-α, IFN-γ), platelet activation (11dTxB2, a metabolite of thromboxane A2, which is synthesized by activated platelets), and oxidative stress (TBARS, 8-OHdG, and 8-isoprostane).
Statistical Analysis
Baseline participant characteristics were summarized using mean (standard deviation [SD]) or median (interquartile range [IQR]) for the continuous variables and frequency (%) for the categorical variables.
Isoflavone intake was determined by the average intake reported at each of 3 visits—or all the visits for which isoflavone data was available. To assess the associations between isoflavones and respiratory morbidity, we conducted generalized estimating equation (GEE) regression with gaussian probability distribution and identity link and exchangeable correlation, regressing the repeated measures of the continuous respiratory outcome on the person-average total isoflavone intake, adjusted by the baseline covariates. For the association of total frequency of exacerbations across the study period with isoflavone intake, negative binomial regression of exacerbations was conducted with the person-average total isoflavone intake, with total follow-up days included as an offset, and adjusted by covariates. For the association between isoflavone and biomarkers, we conducted GEE regression with gaussian probability distribution and identity link and exchangeable correlation regressing the repeated measures of the continuous log-transformed biomarkers on the person-average total isoflavone intake, adjusted by the baseline covariates. All analyses were adjusted by person-average total energy (kcal) and baseline covariates: age, gender, race, educational attainment, household income, smoking pack years, obesity, comorbidity count,18 and FEV1 %pred (except when FEV1 %pred was the outcome). To test the linearity assumption and to illustrate the functional shape of outcomes and total isoflavone more flexibly, a restricted cubic spline curve model was used. As secondary analysis, all analyses were repeated for each individual isoflavone. Based on previous evidence of the effect of omega-3 fatty acids on COPD morbidity,3,19 sensitivity analyses were conducted by further adjusting by omega-3 fatty acid intake (eicosapentaenoic acid [EPA] and docosahexaenoic acid [DHA]). Given results of association of isoflavone with urine thromboxane in current analysis, additional sensitivity analyses were conducted adjusting for antiplatelet use (aspirin or clopidogrel). Lastly, effect modification by EPA+DHA on the association between isoflavone and outcomes were assessed using 2-way interaction models with aforementioned covariate adjustments.
All analyses were conducted with Stata/IC 18.0 software (StataCorp; College Station, Texas); and statistical significance criteria was set at p<0.05.
Results
Study Population Characteristics
Ninety-nine individuals had available FFQ, biomarker, and respiratory outcome data, comprising the entire urban cohort of the CURE COPD study. Participants had a mean age of 66.4 years, 55% were female, and 41% were White (Table 1). Participants had smoked an average of 46.3 pack years and the mean FEV1 %pred was 49.8%. Two-thirds of participants (66%) had an annual household income of less than $30,000, and approximately half of participants (49%) had educational attainment of high school education or less.
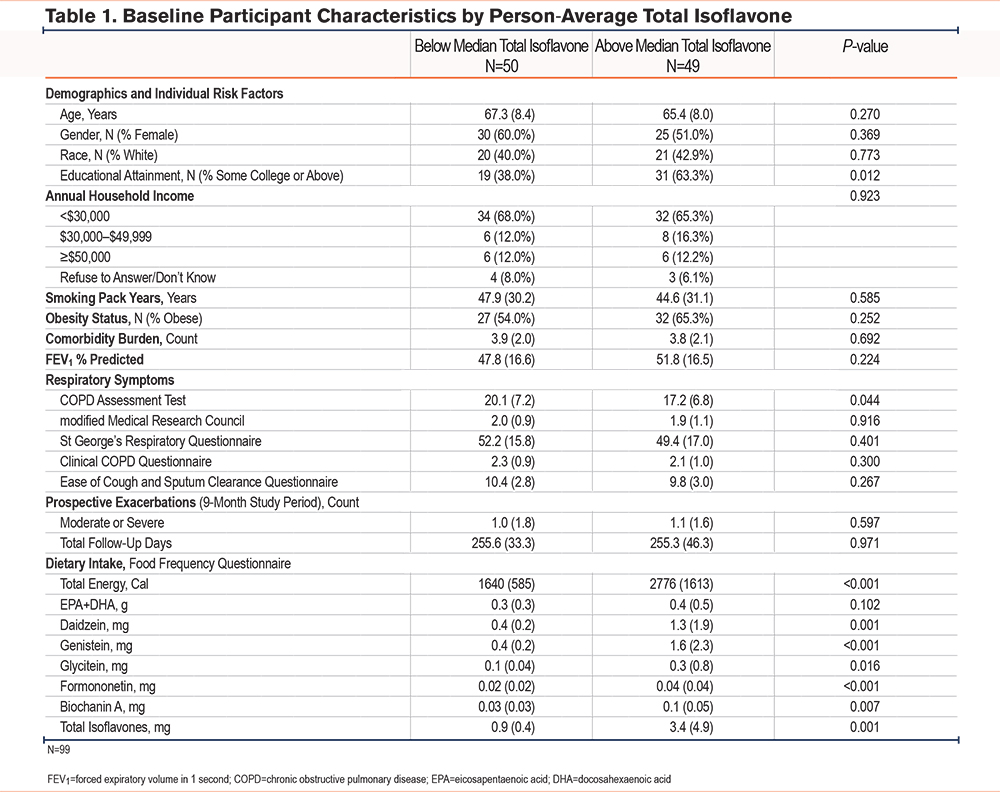
The mean and median total isoflavone intake was 1.8mg (SD=3.1) and 1.0mg (IQR=0.7, 1.7), respectively (Figure 1). Compared to participants with lower isoflavone intakes, those with higher isoflavone intake (above median intake) were significantly more likely to have an educational attainment of at least some college (63% versus 38%) and consumed significantly more calories (2776kcal/day versus 1639.5kcal/day). No other significant demographic differences were noted between individuals with high versus low isoflavone intake.
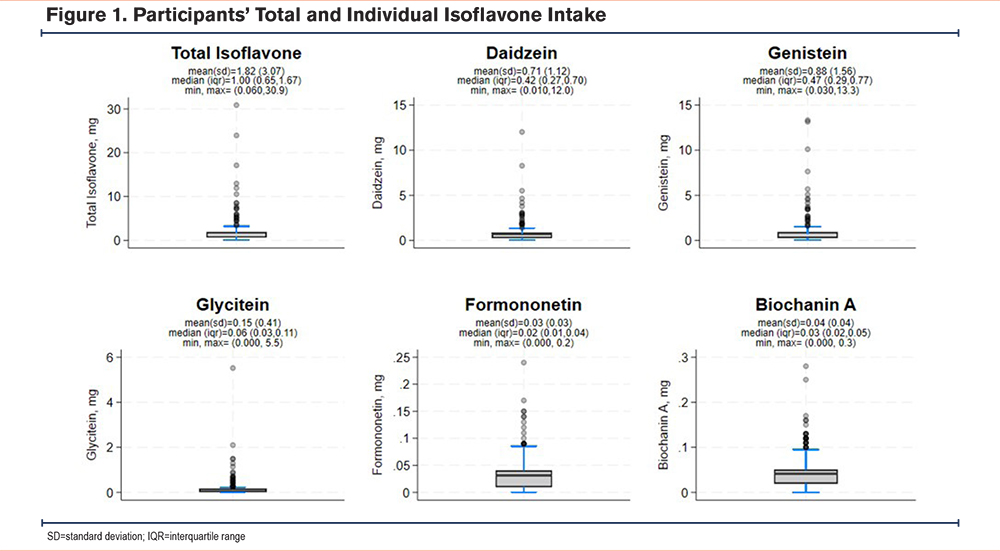
Associations Between Isoflavone Intake and COPD Morbidity
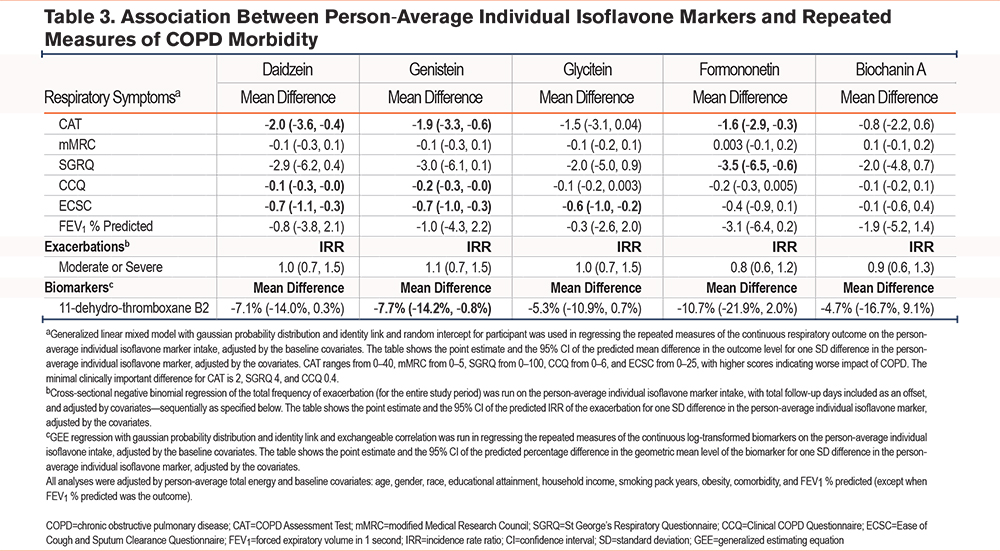
Adjusting for covariates, including educational attainment and caloric intake among other a priori covariates, a higher total isoflavone intake was associated with better respiratory outcomes (Table 2, Figure 2). One SD increase in total isoflavone was associated with a lower CAT score (ß -2.0, P=0.011), a lower CCQ score (ß -0.2, P=0.029), and a lower ECSC score (ß -0.7, P<0.001); and showed a trend towards a better SGRQ score, although this association did not reach statistical significance. There was no observed difference in dyspnea (mMRC) or rates of moderate or severe exacerbations across total isoflavone intake. For each SD increase in total isoflavone there was 7% lower urinary 11dTxB2 concentration (ß -7.4%, P=0.044). No significant association was observed between isoflavone intake and biomarkers of inflammation or oxidative stress.
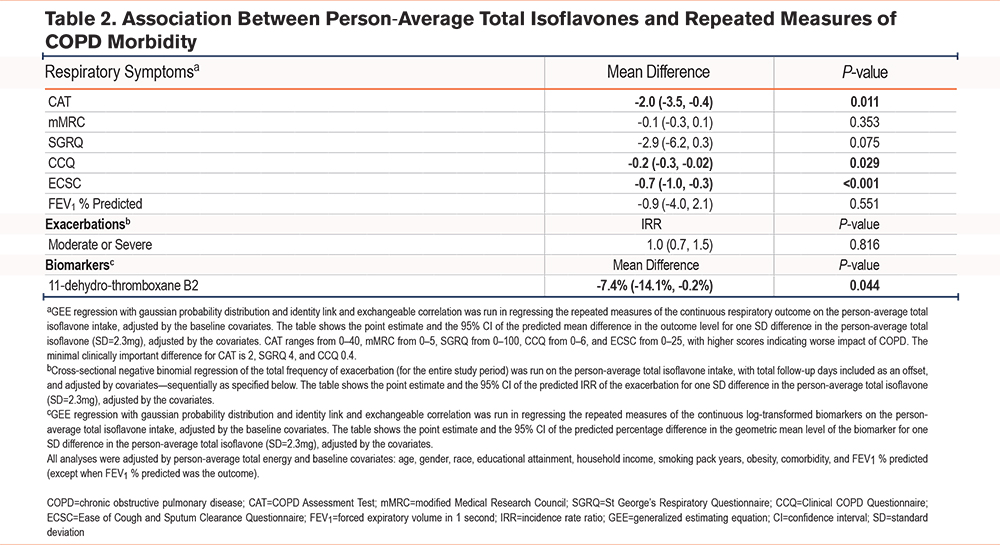
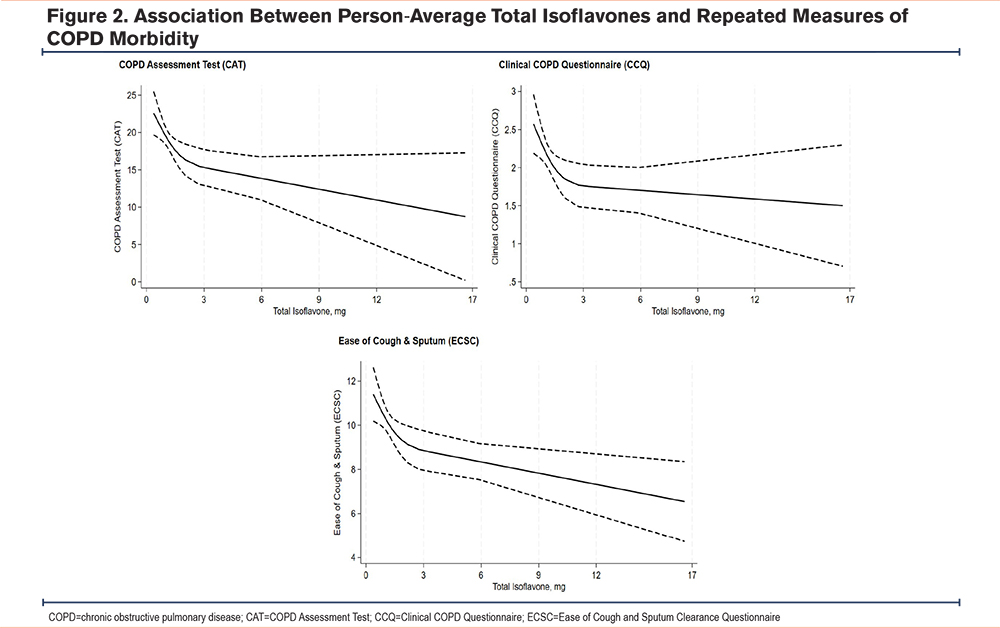
Among individual isoflavones (Table 3), higher intakes of both daidzein and genistein were associated with lower CAT, CCQ, and ECSC scores. Formononetin was associated with lower SGRQ and CAT scores. Glycitein was associated with lower ECSC scores. There was a trend towards association with decreased urinary thromboxane levels for all aforementioned isoflavones, but this association only reached statistical significance for genistein. Biochanin A was not significantly associated with any respiratory outcomes or measured biomarkers.
Sensitivity Analyses
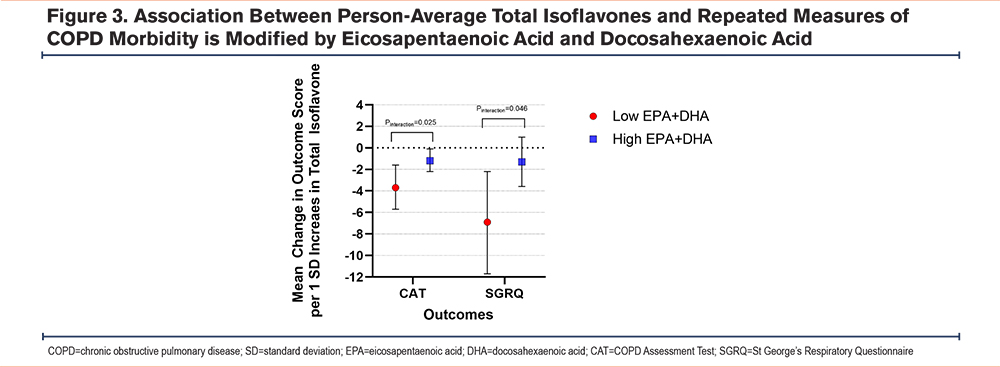
In sensitivity analyses, the association of increased isoflavone intake with better respiratory morbidity was robust to adjustment by omega-3 fatty acid intake. However, there was a significant 2-way interaction between total isoflavones and person-average omega-3 intake (EPA + DHA) for CAT and SGRQ scores, such that the beneficial association of isoflavone with CAT and SGRQ was more marked at lower omega-3 intake (Figure 3).
Given that isoflavone intake was associated with urinary 11dTxB2 concentration, a marker of platelet activation, additional sensitivity analyses were conducted showing that the association of increased isoflavone intake with decreased respiratory morbidity was robust to adjustment by antiplatelet medication (aspirin or clopidogrel) use. The association of total isoflavone intake with urinary 11dTxB2 concentration was somewhat attenuated by adjustment by omega-3 fatty acid intake and by adjustment by antiplatelet medication use. When adjusted by omega-3 fatty acid intake, the association was no longer statistically significant but the effect size remained similar (beta= -6.3%, p=0.16). When adjusted by antiplatelet medication use, the association with 11dTxB2 was no longer statistically significant (beta= -4.2%, p=0.17).
Discussion
This prospective observational study of urban, low-income individuals with COPD demonstrates an association between higher intake of isoflavones and lower respiratory morbidity. Specifically, individuals with higher average intake of isoflavones reported improved health status (CAT, CCQ), and cough and sputum properties (ECSC). There was also a significant interaction between isoflavone and omega-3 fatty acid intake on outcomes, with the beneficial association of isoflavones with health status (CAT) and respiratory-specific quality of life (SGRQ) being more marked for individuals with low omega-3 intake. These findings add to the growing body of literature regarding diet and lung health.
Isoflavones, which can be found in soybean and soybean products, are phytoestrogens – naturally occurring phenolic plant compounds that resemble estrogens.20 Daidzein and genistein account for the bulk of isoflavone intake in humans, with their primary dietary source being soybeans and soybean products. Glycitein is likewise found in soybeans, but in smaller quantities and appears to be less potent biologically. Formononetin and biochanin A are primarily found in red clover – not a typical part of the human diet beyond phytoestrogen supplements – and are converted in vitro into the more potent daidzein and genistein.21 Data from the National Health and Nutrition Examination Survey show that isoflavone intake is higher among individuals with higher family income.22 Our cohort represents a relatively lower income population and isoflavone intake among participants in our cohort was slightly lower than the national average for all individuals and similar to intake among individuals with family income levels under 131% of the federal poverty level.23
Our results showed an association between higher total isoflavone intake and better respiratory outcomes, including health status as assessed by the CAT and CCQ as well as cough symptoms as assessed by the ECSC. There was additionally a trend towards better respiratory-specific quality of life as assessed by the SGRQ, suggesting that a larger study may have had sufficient power to detect a statistically significant difference. Of these findings, the mean difference in the CAT reached the established minimal clinically important difference (MCID) of 2.0. The mean difference in the CCQ of 0.2 did not reach the established MCID of 0.4, and there is no established MCID for the ECSC. Because the mean isoflavone intake in the study population was lower than the national mean (1.8mg versus 2.35mg per day),22 it is possible that larger effect sizes could be seen in a population with a higher mean or larger range of isoflavone consumption.
The association between improved CAT, CCQ, and ECSC scores was specifically associated with higher intake of the individual isoflavones daidzein and genistein. This finding is consistent with prior work that has demonstrated that these isoflavones are the most biologically active and account for the large majority of human isoflavone consumption.20 These results were independent of caloric intake and other dietary measures shown to be associated with improved COPD morbidity, such as omega-3 polyunsaturated fatty acid consumption. Further, our results suggest that the benefit of eating higher isoflavone rich foods may be greater in populations that have otherwise lower quality nutritional intake, such as lower omega-3 intake.
There are several potential mechanisms by which isoflavone intake may impact respiratory health. One proposed mechanism is through the antioxidant and anti-inflammatory effects of isoflavones,24 which could reduce the chronic inflammatory state that is implicated in COPD progression and morbidity,6 though our study did not find an association between increased isoflavone intake and decreased circulating inflammatory markers. An additional potential mechanism is through the downregulation of platelet activation by isoflavones. Platelet activation is elevated in COPD and has been associated with respiratory morbidity.4,25 Both genistein and daidzein are known to downregulate platelet activation26,27 by specifically blocking synthesis of thromboxane A2, the precursor of 11dTxB2. This mechanism is further supported by our finding that increased isoflavone intake is associated with decreased systemic platelet activation as measured by urinary 11dTxB2.
Notably, isoflavone intake was not significantly associated with the mMRC or prospective exacerbation rates. The mean difference in the mMRC was towards improvement in dyspnea, so it is possible that the current study was underpowered to detect a significant difference. On the other hand, since the mMRC is a measure specifically of dyspnea, the proposed anti-inflammatory and antiplatelet mechanisms of isoflavone intake may impact respiratory morbidity primarily through improved of cough and sputum production rather than through dyspnea. Regarding exacerbations, it is possible that the effect of isoflavone intake on respiratory morbidity can modify day-to-day symptoms, but that the associated anti-inflammatory and antiplatelet effects are too modest to modify the course of an exacerbation triggered by a respiratory infection or significant environmental exposure.
While interesting, these results should be interpreted with caution given the experience of isoflavone supplementation in asthma. The isoflavone genistein has been found to attenuate bronchoconstriction due to antigen challenge of airways both in vitro and in vivo in a Guinea-Pig model of asthma.28-30 Further, 2 observational studies demonstrated an association between dietary intake of the isoflavone genistein with improved lung function and asthma control.31,32 Based on this compelling evidence, a randomized, double-blind trial of isoflavone supplementation versus placebo was carried out across centers in the American Lung Association Asthma Clinical Research Centers network. While mean plasma genistein levels increased in the intervention group, there was no significant difference in FEV1 or markers of asthma control after 6 months.33 In spite of this finding, there are notable differences between asthma and COPD which suggest that isoflavone intake may still benefit respiratory health in COPD. Preclinical evidence for the effect of isoflavones in asthma focuses on acute bronchoconstriction and eosinophilic inflammation, which may play less of a role in COPD than in asthma. Furthermore, while platelets activation has also been implicated in the pathophysiology of asthma, it may have a greater influence in COPD.
Among the strengths of this study is the extensive clinical and nutritional data collected on participants. This allows the exploration of the differential associations of various nutrients such as isoflavones and omega-3 fatty acids to respiratory health. Furthermore, the study population is enriched for low-income individuals living in an urban environment and accordingly is representative of the specific challenges of such an environment, including higher exposure to stressors on the respiratory system including indoor and outdoor air pollution and decreased access to potential mediators such as affordable healthy foods, which may result in more pulmonary inflammation and oxidative stress. Regarding potential limitations of this study, it is possible that the increased intake of isoflavone-rich foods such as tofu and soy products is indicative of a more health-conscious diet that could be associated with improved health habits overall; however, even in Japanese cohorts with generally higher intake of soy products and fish, soy intake has been positively associated with improved respiratory symptoms in COPD.34,35 Second, measurement errors in dietary self-assessment may be increased for tofu and soy products that are not widely consumed in Western populations; however, the FFQ used has been shown to correlate well with urinary excretion of isoflavone metabolites.36 Our biomarker analyses were limited by our small sample size and the general lack of sensitive and specific serum biomarkers in COPD, and we did not perform adjustment for multiple testing.37 Finally, as noted above, our small sample size may have limited our ability to detect significant differences in the SGRQ and mMRC.
Conclusion
This study of former smokers with COPD living in urban Baltimore, Maryland found a significant association between increased isoflavone intake and improved respiratory morbidity. Individuals with higher isoflavone intake reported improved health status and cough symptoms. Additionally, higher isoflavone intake was associated with lower systemic platelet activation. These findings suggest that isoflavone intake and associated downregulation of platelet activation may be one mechanism by which diet influences COPD morbidity.
Acknowledgements
Author contributions: NH conceptualized and designed the study. HW conducted statistical analysis. DB and EQ drafted the manuscript. All authors contributed to the interpretation of the findings, critically revised the manuscript, and approved the final version for submission.
EPA Statement: This publication was developed under Assistance Agreement No.83615001 awarded by the U.S. Environmental Protection Agency to Nadia Hansel. It has not been formally reviewed by the EPA. The views expressed in this document are solely those of Daniel C. Belz, MD, MPH, Ernesto Quiroz, Han Woo, PhD, Mariah K. Jackson, PhD, RD, Nirupama Putcha, MD, MHS, Ashraf Fawzy, MD, MPH, Wendy Lorizio, MD, MPH, Corrine K. Hanson, PhD, RD, and Nadia N. Hansel, MD, MPH, and do not necessarily reflect those of the Agency. The EPA does not endorse any products or commercial services mentioned in this publication.
Declarations of Interest
The authors have no conflicts of interest to declare.